Electron gain enthalpy is the enthalpy change that occurs when an electron is added to an isolated gaseous atom to form a negative ion. It helps explain the tendency of elements to accept electrons and plays an important role in understanding periodic properties and chemical reactivity. The study of electron gain enthalpy includes its definition, units, factors affecting it, periodic trends, and successive electron gain enthalpies.
What is Electron Gain Enthalpy ?
Just as energy is required to remove an electron from an atom, energy is released when an electron is added to a neutral atom. This is called electron gain enthalpy.
Thus, electron gain enthalpy is the energy released when electron is added to an isolated gaseous atom to form the gaseous negative ion.
What is the definition of Electron Gain Enthalpy ?
Electron gain enthalpy is defined as the energy change that occurs when an electron is added to an isolated gaseous atom to form a gaseous negative ion. It is represented as ΔegH and expressed in kilojoules per mole (kJ mol⁻¹). $$A(g) + e^{-}(g) \;\longrightarrow\; A^{-}(g) \quad \Delta H = \Delta_{\text{eg}}H$$
What happens to electron gain enthalpy when addition of an electron releases energy?
When the addition of an electron to a neutral atom releases energy, the Electron Gain Enthalpy ($\Delta_{eg}H$) is negative.
In chemistry and thermodynamics, the sign convention is based on the system (the atom). If the atom loses or releases energy to the surroundings, that process is exothermic, and the enthalpy change is represented with a negative sign. Energy is released when the incoming electron is strongly attracted to the nucleus. This usually happens when an atom has strong affinity to fill its valence shell to reach a more stable, lower-energy state (like a noble gas configuration).
As the definition implies, the magnitude of the electron gain enthalpy measures the ability of an atom to hold an additional electron. If an atom has more tendency to accept an electron, large energy will be released. Consequently, electron gain enthalpy will be highly negative.
On the other hand, if an atom has less tendency to hold the electron, small amount of energy will be released, leading to a small negative value of electron gain enthalpy.
High Attraction = More Energy Released = More Negative $\Delta_{eg}H$.
It is important to distinguish between “Electron Affinity” and “Electron Gain Enthalpy,” as their signs are often opposite :
Electron Gain Enthalpy ($\Delta_{eg}H$) : Measures the heat change. If energy is released, it is negative.
Electron Affinity (EA) : Measures the “love” for electrons. If energy is released, the affinity is considered positive.
But the Electron Gain Enthalpy process may be exothermic (energy released) or endothermic (energy absorbed).
| Process | Energy Direction | Sign of ΔegH | Example |
| Exothermic | Energy is Released | Negative ( < 0 ) | Halogens (Cl, F) |
| Endothermic | Energy is Absorbed | Positive ( > 0 ) | Noble Gases, Alkaline Earth Metals |
What does a highly negative electron gain enthalpy indicate?
A highly negative value of Electron Gain Enthalpy indicates that the atom has a strong tendency to accept an electron, releasing a large amount of energy.
What does a small negative or positive electron gain enthalpy indicate?
A small negative (or positive) value of Electron Gain Enthalpy indicates a weaker tendency to accept an electron, releasing less energy (or requiring energy input).
What are the common units of Electron Gain Enthalpy ?
The electron gain enthalpy is expressed in units of kJ mol–1 or electron volt (eV) per atom or kcal mol–1.
What is the conversion relation between electron volt and kJ/mol?
The conversion relation between electron volt and kJ/mol are in the following table.
| Unit | Symbol | Conversion |
|---|---|---|
| Kilojoule per mole | kJ mol⁻¹ | – |
| Electron volt per atom | eV/atom | 1 eV per atom = 96.49 kJ/mol |
| Kilocalorie per mole | kcal mol⁻¹ | 1 eV per atom = 23.06 kcal/mol |
For example in chlorine atom :
Cl (g) + e− → Cl–(g)
Electron gain enthalpy = -349 kJ/mol
How do we convert electron gain enthalpy released per atom into energy released per mole?
Multiply the energy released per atom by Avogadro’s number (6.022 × 10²³ atoms mol⁻¹).
How do we convert from kJ mol⁻¹ to eV per atom?
Use the relation: 1 eV atom−1 = 1.602 x 10-19 x 6.022 x 1023 x 10-3= 96.49 kJ mol−1
So, divide the value in kJ mol⁻¹ by 96.49.
Why do noble gases (group 18) have positive electron gain enthalpy values?
Some elements, such as noble gases, have large positive electron gain enthalpy values. They already possess stable electronic configurations and have no tendency to accept additional electrons. If forced to gain an electron, it must enter a higher principal quantum shell, resulting in an unstable arrangement. So that’s why noble gases, have large positive electron gain enthalpy values as shown in following table.
| Element | Atomic Number (Z) | ΔegH (kJ mol⁻¹) |
| He (Helium) | 2 | + 48 |
| Ne (Neon) | 10 | + 116 |
| Ar (Argon) | 18 | + 96 |
| Kr (Krypton) | 36 | + 96 |
| Xe (Xenon) | 54 | + 77 |
| Rn (Radon) | 86 | + 68 |
What are the factors on which Electron Gain Enthalpy Depends ?
Electron gain enthalpy is the amount of energy change when an atom in the gaseous state accepts an extra electron to form a negative ion. Its value is influenced by several factors :
(i) Nuclear Charge
The greater the nuclear charge, the stronger will be the attraction between the positively charged nucleus and the incoming electron. A stronger attraction means more energy is released when the electron is added, making the electron gain enthalpy more negative.
Example :
Fluorine (Z = 9) has a high nuclear charge for its small size, hence it has a highly negative electron gain enthalpy.
Sodium (Z = 11) has lower effective nuclear charge due to more inner electrons shielding the nucleus, so its electron gain enthalpy is less negative.
“Learn more about Nuclear Charge“
(ii) Size of the Atom
As the atomic radius increases, the distance between the nucleus and the incoming electron also increases. A larger distance reduces the electrostatic attraction, so less energy is released when the electron is gained. Hence, electron gain enthalpy becomes less negative with increasing size of the atom.
Example:
Chlorine (Cl) has a smaller atomic size compared to iodine (I). Therefore, chlorine has a more negative electron gain enthalpy than iodine.
“Learn more about Atomic Radius Trends“
(iii) Electronic Configuration of Atom
Atoms with stable electronic configurations (such as noble gases, half-filled, or fully filled subshells) show very little tendency to gain electrons.
In such cases, either : Energy has to be supplied to force an electron into the stable configuration (making ΔegH positive), or the energy released is very small (making ΔegH only slightly negative).
Examples :
Noble gases (He, Ne, Ar) have completely filled shells, so they resist gaining electrons and hence their ΔegH is positive.
Nitrogen (1s² 2s² 2p³) has a half-filled stable 2p subshell; hence its electron gain enthalpy is less negative than oxygen, even though oxygen has a smaller size.
(iv) Shielding Effect (Screening Effect)
The electrons present in the inner shells repel the incoming electron and shield it from the full attraction of the nucleus. Higher the number of inner-shell electrons, greater will be the shielding, and thus weaker will be the effective nuclear attraction felt by the incoming electron. As a result, the electron gain enthalpy becomes less negative.
“Learn more about Shielding Effect (Screening Effect)“
Example :
In halogens, fluorine and chlorine have similar nuclear charges, but in chlorine, the added electron experiences slightly less repulsion (less crowding of electrons) compared to fluorine. That’s why chlorine actually has a more negative electron gain enthalpy than fluorine.
Why do electron gain enthalpy values of noble gases are positive while those of Beryllium (Be), Magnesium (Mg), Nitrogen (N) and Phosphorus (P) are almost zero ?
The electron gain enthalpy values of noble gases are positive. This is because they have stable electronic configuration of ns2 np6 and thus they have absolutely no tendency to take an additional electron. This means that the incoming electron enters the next higher principal quantum level and does not feel any attraction for the nucleus. Thus, energy is required to force the electron in their atoms and therefore, their electron gain enthalpies are positive.
Similarly, the low (almost zero) electron gain enthalpy values for Be, Mg, N and P can be explained due to the extra stability of completely filled 2s-orbitals and 3s-orbitals of valence electrons in Be (2s2 ) and Mg (3s2 ) respectively and of half-filled 2p3-and 3p3-orbitals in N and P respectively. Therefore, the configurations show little tendency to gain any electron and hence their electron gain enthalpies are very low (almost zero).
Why do halogens (group 17) have highly negative electron gain enthalpy values ?
The electron gain enthalpies of the halogens (group 17 elements) are highly negative. This is due to the fact that halogens have the general electronic configuration of ns2np5 and have only one electron less than the stable noble gas (ns2np6) configurations. Thus, they have very strong tendency to accept an additional electron and their electron gain enthalpies are, therefore, highly negative.
What is the general trend of Electron Gain Enthalpy across a period and down a group ?
Due to the lack of sufficient data, the changing trends in electron gain enthalpies on moving down a group along the period in the periodic table are less well defined than those for ionisation enthalpies. However, it has been observed in Table shown of some elements that the electron gain enthalpy in kJ mol⁻¹, in general, becomes more negative from left to right in a period and becomes less negative as we go from top to bottom in a group.
| Atom | H | He | Li | Be | B | C | N | O | F | Ne |
|---|---|---|---|---|---|---|---|---|---|---|
| ΔegH | –73 | +48 | –60 | ≈0 | –83 | –127 | ≈0 | –141 | –328 | +116 |
| Atom | Na | Mg | Al | Si | P | S | Cl | Ar |
|---|---|---|---|---|---|---|---|---|
| ΔegH | –53 | ≈0 | –50 | –119 | –74 | –200 | –349 | +97 |
| Atom | K | Ca | Ga | Ge | As | Se | Br | Kr |
|---|---|---|---|---|---|---|---|---|
| ΔegH | –48 | –2 | –36 | –116 | –77 | –195 | –325 | +96 |
| Atom | Rb | Sr | Te | I | Xe |
|---|---|---|---|---|---|
| ΔegH | –47 | –5 | –190 | –295 | +77 |
| Atom | Cs | Po | At | Rn |
|---|---|---|---|---|
| ΔegH | –46 | –174 | –270 | +68 |
Electron gain enthalpy refers to the energy change that occurs when an electron is added to a neutral gaseous atom. Its trend in the periodic table can be summarized in the next sections.
“Also Learn About Atomic Radius Trends“
How does Electron Gain Enthalpy vary along a Period?
As we move across a period (left to right), the atomic size decreases while the nuclear charge increases. Both these factors enhance the attraction between the nucleus and the incoming electron. Consequently, the electron gain enthalpy becomes more negative across a period.
However, certain irregularities are observed in the general trend. These are mainly due to the stable electronic configurations of certain atoms.
Why Fluorine (F) has a more negative electron gain enthalpy than Oxygen (O) ?
The Electronic Configurations of Oxygen is 1s22s22p4 and Fluorine is 1s22s22p5. Fluorine has only one electron less than the stable noble gas configuration (2s²2p⁶), while oxygen has two electrons less. The important points regarding fluorine are as :-
Closer to noble gas configuration : Fluorine needs just one electron to achieve the stable octet. This makes the electron gain more favorable, so more energy is released.
Smaller size : Fluorine is smaller than oxygen, so the nucleus exerts a stronger attraction on the added electron.
Stronger effective nuclear charge (Zeff) : Fluorine’s nucleus attracts the incoming electron more strongly than oxygen’s, releasing more energy.
That’s why F has a more negative electron gain enthalpy than O.
How does Electron Gain Enthalpy vary Down a Group?
On moving down a group, the size and nuclear charge increases. But the effect of increase in atomic size is much more pronounced than that of nuclear charge and thus the additional electron feels less attraction by the large atom. Consequently, electron gain enthalpy becomes less negative. This is clear from decrease of electron gain enthalpy in going from chlorine (Cl) to bromine (Br) and to iodine (I).
“Learn More About Ionization Enthalpy Trends“
Why does chlorine have more negative electron gain enthalpy than fluorine despite fluorine being smaller?
The electron gain enthalpy, in general, becomes less negative from top to bottom in a group. However, it is observed that fluorine (F) atom has unexpectedly less negative electron gain enthalpy than chlorine (Cl) atom. The electron gain enthalpy values for the halogens are shown in Figure below.
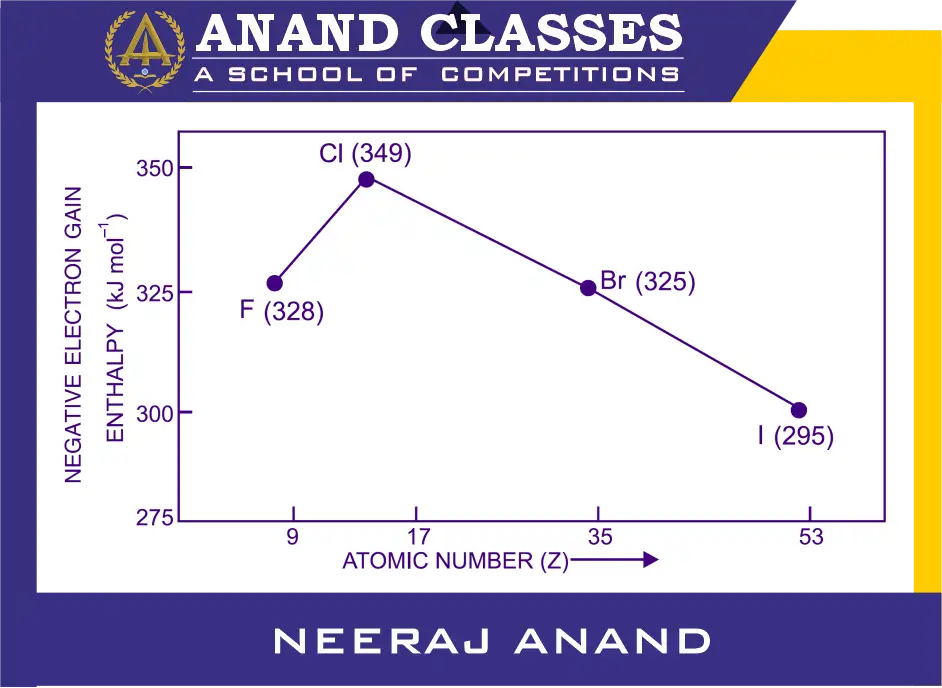
The less negative electron gain enthalpy value of fluorine (F) atom is due to the exceptionally very small size of fluorine (F) atom (atomic radius ≈ 42 pm). As a consequence of exceptionally small size, there are strong inter electronic repulsions are present due to crowding of 7-electrons in the relatively compact 2p-subshell of fluorine and thus the incoming electron does not feel much attraction. When an extra electron enters, it experiences very high electron–electron repulsions. This reduces the stability of fluorine (F) atom gained after accepting the electron.
In chlorine, the incoming electron enters the larger 3p orbital, where repulsions are less significant. The added electron is more easily accommodated, giving greater stability. Thus chlorine has more tendency to attract incoming electron than fluorine. Therefore, chlorine releases more energy on accepting an electron and hence ΔegH of chlorine is more negative (–349 kJ mol⁻¹) compared to fluorine (–328 kJ mol⁻¹).
Hence, the element with most negative electron gain enthalpy is chlorine.
Why does electron gain enthalpy of halogens (group 17) follows the order F < Cl > Br > I
Halogens have a strong tendency to gain one electron because by accepting an electron they attain a stable noble gas configuration. Due to this reason, the electron gain enthalpy values of halogens are highly negative.
The order of electron gain enthalpy among halogens is :
$$
\text{Cl} > \text{F} > \text{Br} > \text{I}
$$
Chlorine has the most negative electron gain enthalpy. Although fluorine is the most electronegative element, its electron gain enthalpy is slightly less negative than chlorine.
This happens because fluorine has a very small atomic size. The incoming electron has to enter the compact $2p$ orbital where electron–electron repulsion is quite high. As a result, less energy is released when fluorine gains an electron.
In chlorine, the additional electron enters the comparatively larger $3p$ orbital. The repulsion between electrons is less, so more energy is released. Therefore, chlorine shows more negative electron gain enthalpy than fluorine.
As we move down the group from chlorine to iodine, atomic size increases and the attraction between the nucleus and the incoming electron decreases. Hence, the tendency to gain an electron decreases and electron gain enthalpy becomes less negative down the group. These values are shown in following table
| Element | ΔegH (kJ mol⁻¹) | Observation |
| F (Fluorine) | – 328 | Smaller size, high inter-electronic repulsion. |
| Cl (Chlorine) | – 349 | Highest (most negative) value in the periodic table. |
| Br (Bromine) | – 325 | Decreases as size increases. |
| I (Iodine) | – 295 | Most loosely held among halogens. |
| Key Point |
|---|
| The order of Electron Gain Enthalpy of halogens is F < Cl > Br > I |
Why is the electron gain enthalpy of sulfur more negative than that of oxygen?
Oxygen has a very small size, so when an extra electron enters, it faces high repulsion in the compact electron cloud. Sulfur, being larger, offers more space, so the added electron experiences less repulsion and more effective attraction by the nucleus. Therefore, electron gain enthalpy of sulfur (–200 kJ mol⁻¹) is more negative than oxygen (–141 kJ mol⁻¹).
It may be noted that, in general, the electron gain enthalpies for some third period elements (e.g. P, S, Cl) are more negative than the corresponding second period members (e.g. N, O, F). This is due to the smaller size of the atoms of the second period elements which would produce larger electron-electron repulsions for the additional electron. This effect is much more pronounced in the smallest fluorine atom as discussed above.
What is meant by Successive Electron Gain Enthalpies and why second and higher electron gain enthalpies are always positive ?
Like ionization enthalpies, the second and higher electron gain enthalpies are also possible. The first electron gain enthalpy involves adding an electron to a neutral atom. Since the atom is neutral, the incoming electron experiences an overall attraction from the nucleus. As a result, in most cases, energy is released (exothermic process) when the first electron is added.
However, the second electron is added to a negatively charged ion and the addition is opposed by coulombic repulsions. The energy has to be supplied to force the second electron into the anion by overcoming the repulsions between negatively charged ion and incoming electron. Hence, second electron gain enthalpy (and higher values) are always positive (endothermic process).
Why is the first electron gain enthalpy of oxygen negative while the second electron gain enthalpy is highly positive?
The first electron gain enthalpy involves adding an electron to a neutral oxygen atom (O). Since the oxygen atom is neutral, the incoming electron experiences an overall attraction from the nucleus. As a result, energy is released when the first electron is added to oxygen atom (O) to form oxygen anion (O–).
First electron gain enthalpy
$$O (g) + e^– \longrightarrow O^– (g); \Delta_{eg}H_1 = – 141 \text{ kJ mol}^{–1}$$
Here, energy is released (negative value) because the nucleus attracts the incoming electron.
However, when the second electron is added to a negatively charged oxygen anion (O–) and the addition is opposed by coulombic repulsions. The energy has to be supplied to force the second electron into the oxygen anion by overcoming the repulsions between negatively charged ion and incoming electron. Hence, second electron gain enthalpy of oxygen is always positive.
Second electron gain enthalpy
$$O^– (g) + e^– \longrightarrow O^{2–}(g) \Delta_{eg}H_2 = + 780 \text{ kJ mol}^{–1}$$
Here, a very high amount of energy must be supplied to push an electron into an already negatively charged ion. Thus, oxygen can easily form O− but forming O2− in the gaseous state is very difficult due to high repulsion.
In other words, the second (and higher) electron gain enthalpies would have positive values. For example, the first and second electron gain enthalpy values of O, S and Se are given below.
| Element | $\Delta_{eg}H_1$ (kJ mol⁻¹) | $\Delta_{eg}H_2$ (kJ mol⁻¹) |
| O | – 141 | + 780 |
| S | – 200 | + 590 |
| Se | – 195 | + 420 |
Why does oxygen not readily form O2− ions in the gaseous state ?
In the gaseous state, forming O2− requires a huge energy input (+780 kJ mol⁻¹ for $\Delta_{eg}H_2$). However, in ionic compounds (like MgO, CaO), this energy is compensated by the high lattice enthalpy of the crystal. Hence, O2− is stable in solids, but difficult to form in the gaseous state.
Compare the second electron gain enthalpies values of oxygen, sulfur, and selenium and why it decreases down the group.
The second electron gain enthalpies values of oxygen, sulfur, and selenium are as follows :-
Oxygen : +780 kJ mol⁻¹
Sulfur : +590 kJ mol⁻¹
Selenium : +420 kJ mol⁻¹
The trend shows that second electron gain enthalpies decreases down the group, because larger atomic size reduces repulsion between the incoming electron and the anion and requiring less energy for addition of incoming electron.
Why third period elements (P, S, Cl) have more negative electron gain enthalpies than second period (N, O, F) Elements ?
In many cases, the third-period elements (P, S, Cl) have more negative electron gain enthalpies than their corresponding second-period elements (N, O, F).
For example :
Nitrogen (N) : –7 kJ/mol (almost zero, since its half-filled p3 is stable)
Phosphorus (P) : –48 kJ/mol (more negative than N)
Oxygen (O) : –141 kJ/mol
Sulphur (S) : –200 kJ/mol (more negative than O)
Fluorine (F) : –328 kJ/mol
Chlorine (Cl) : –349 kJ/mol (more negative than F)
Second-period atoms (N, O, F) are extremely small in size. When an additional electron is added, it has to enter a very compact 2p orbital. This compact orbital is already crowded with electrons and the new electron faces strong electron–electron repulsions. Due to this repulsion, the attraction of the nucleus is not felt strongly, and hence less energy is released (ΔegH is less negative).
On the other hand the third-period atoms (P, S, Cl) are larger in size compared to second-period atoms. The incoming electron enters the 3p orbital, which is more diffused and spacious. As a result the repulsion between electrons is less. So the incoming electron can be accommodated more easily. Hence the nucleus can exert a stronger net attraction and more energy is released so that ΔegH becomes more negative.
Special Case : Fluorine
Fluorine is the smallest atom in the group. Its 2p orbital is extremely compact. Adding an extra electron here produces very high inter-electronic repulsion, more than in any other element. That’s why the anomaly is most pronounced in fluorine, whose electron gain enthalpy is less negative than chlorine, despite fluorine being more electronegative.
Among Phosphorus (P), Sulphur (S), Chlorine (Cl), Fluorine (F) Elements will have the most negative electron gain enthalpy and which the least negative?
The given elements can be placed in the periodic table as follows :
| Group No. | 15 | 16 | 17 |
|---|---|---|---|
| 2nd Period | – | – | F |
| 3rd Period | P | S | Cl |
Across a Period (Left to Right) : Electron gain enthalpy becomes more negative as we move across a period from left to right.
Down a Group (Top to Bottom) : Electron gain enthalpy becomes less negative as we move down a group because atomic size increases and the added electron experiences less attraction from the nucleus.
Phosphorus (P) : Being in Group 15, it has relatively stable half-filled (3p3) orbitals. Thus, it resists gaining an extra electron. Hence, it has the least negative electron gain enthalpy.
Fluorine (F) : Although it is very electronegative, the incoming electron has to enter the small 2p orbital where strong inter-electronic repulsions occur. This makes its electron gain enthalpy less negative than expected.
Chlorine (Cl) : The incoming electron enters the larger 3p orbital, where repulsions are comparatively less. As a result, Cl has more negative electron gain enthalpy than F.
Hence Chlorine (Cl) has the most negative electron gain enthalpy while Phosphorus (P) has the least negative electron gain enthalpy.
What are the differences between electron gain enthalpy and ionization enthalpy?
While both Ionization Enthalpy and Electron Gain Enthalpy measure energy changes related to electrons, they represent opposite physical processes. One describes how hard an atom “clings” to its electrons, while the other describes how much an atom “wants” a new one.
“Learn More Ionization Enthalpy“
| Feature | Ionization Enthalpy (ΔiH) | Electron Gain Enthalpy (ΔegH) |
| Basic Definition | The minimum energy required to remove the most loosely bound electron from an isolated gaseous atom. | The enthalpy change that occurs when an electron is added to an isolated gaseous atom. |
| Process | $X(g) + \text{Energy} \rightarrow X^+(g) + e^-$ | $X(g) + e^- \rightarrow X^-(g) \pm \text{Energy}$ |
| Nature of Energy | It is always an Endothermic process (energy is absorbed). | It is usually Exothermic (energy released), but can be Endothermic for noble gases. |
| Sign of $\Delta H$ | The value is always Positive ($+$). | The value can be Negative ($-$) or Positive ($+$). |
| Force Involved | Overcomes the attraction between the nucleus and the electron. | Results from the attraction between the incoming electron and the nucleus. |
| Resulting Ion | Leads to the formation of a Cation (positive ion). | Leads to the formation of an Anion (negative ion). |
| Successive Values | Successive values ($IE_2, IE_3$) always increase as it becomes harder to remove electrons. | Successive values ($\Delta_{eg}H_2, \Delta_{eg}H_3$) are always positive due to inter-electronic repulsion. |
Solved Numerical Examples Based on Electron Gain Enthalpy
Solved numerical examples based on electron gain enthalpy help students understand how energy changes occur when an electron is added to an isolated gaseous atom. These problems involve concepts such as sign convention, energy calculations, electron affinity trends, and interpretation of electron gain enthalpy values for different elements. Practicing numerical problems strengthens conceptual understanding and improves problem-solving skills for board and competitive examinations.
Q. Electron gain enthalpy of chlorine is 349 kJ mol⁻¹. How much energy (in kJ) is released when 1 g of chlorine is completely converted to Cl– ions in the gaseous state?
Solution :
Cl (g) + e– (g) → Cl– (g) ΔegH = −349 kJ/mol
This means that 349 kJ energy is released when 1 mole (35.5 g) of chlorine atoms gains electrons.
So the energy released for 1 g of chlorine = 349/35.5 x 1 = 9.83 kJ
Hence when 1 g of chlorine is completely converted to Cl– ions, the energy released is 9.83 kJ
Q : The amount of energy released when one million atoms of iodine in vapour state are converted to I– ions is 4.9 × 10-13 J according to the reaction: I(g) + e– → I–(g). Express the electron gain enthalpy of iodine in: (i) kJ mol⁻¹ (ii) eV per atom
Solution :
Energy released for one million (1 × 106) atoms of iodine = 4.9×10−13 J
Energy released for 1 atom of iodine = 4.9 × 10−13/1 × 106 = 4.9 × 10−19J
Therefore energy released for 1 mole (6.02 × 10²³ atoms) = 4.9 × 10−19 × 6.022 × 1023 = 2.95 × 105 J
Thus, the electron gain enthalpy of iodine = –295 kJ mol⁻¹.
We know 1 eV/atom = 96.49 kJ/mol
Electron gain enthalpy in eV/atom = 295/96.49 =3.06 eV/atom
Electron Gain Enthalpy Conceptual Questions and Answers – Class 11 Chemistry JEE NEET
What is electron gain enthalpy?
Electron gain enthalpy is the amount of energy released or absorbed (usually expressed in kJ/mol) when an electron is added to an isolated gaseous atom to form a negatively charged ion (anion). If energy is released then electron gain enthalpy is negative and if energy is absorbed then electron gain enthalpy is positive.
Why is electron gain enthalpy usually negative?
Most atoms have a tendency to gain extra electrons to achieve stable electronic configurations (like noble gases). Since attraction between the nucleus and the added electron releases energy, electron gain enthalpy is generally negative.
What is successive electron gain enthalpy? Why is the second electron gain enthalpy positive?
First electron gain enthalpy : Energy released when the first electron is added to a neutral atom.
Second electron gain enthalpy : Energy change when an electron is added to a negatively charged ion.
Since a negative ion strongly repels an incoming electron, energy must be supplied. Hence, the second electron gain enthalpy is always positive.
How is electron affinity different from electron gain enthalpy?
Electron Affinity (EA) : It is the energy released when an electron is added to an atom, expressed as a positive quantity.
Electron Gain Enthalpy (ΔegH) : It is the actual energy change, which may be negative or positive.
Why is electron gain enthalpy of chlorine more negative than that of fluorine, even though fluorine is smaller in size?
Normally, a smaller size means stronger attraction between the nucleus and the incoming electron, so ΔegH should be more negative. But in fluorine, the atomic size is very small, and the incoming electron has to enter the compact 2p orbital, where strong electron–electron repulsions are present due to crowding of electrons. In chlorine, the incoming electron enters the larger 3p orbital, where repulsions are less significant. Therefore, chlorine releases more energy on accepting an electron. Hence ΔegH of chlorine is more negative (–349 kJ mol⁻¹) compared to fluorine (–328 kJ mol⁻¹).
Why do noble gases have positive electron gain enthalpy?
Noble gases (He, Ne, Ar, etc.) have completely filled electronic configurations (ns² np⁶). This configuration is very stable, and adding an electron would require placing it in the next higher energy orbital (new shell). Instead of releasing energy, the atom would need extra energy input to force the electron in. Thus, electron gain enthalpy of noble gases is positive.
Why is the electron gain enthalpy of nitrogen less negative than that of oxygen?
Nitrogen (1s² 2s² 2p³) has a half-filled stable p-orbital configuration. Adding an electron would disturb this stability, so nitrogen has very low tendency to accept an electron, its ΔegH is less negative. Oxygen (1s² 2s² 2p⁴) does not have a half-filled stable configuration, so it can accept an electron more easily to form O⁻ with 2p⁵. Hence, oxygen has a more negative electron gain enthalpy than nitrogen.
Why does electron gain enthalpy become less negative down a group?
As we move down a group, the atomic size increases and shielding effect increases. The nucleus is farther from the incoming electron, and inner electrons reduce the effective nuclear attraction. As a result, less energy is released when an extra electron is added. Hence, ΔegH becomes less negative down the group.
Why does electron gain enthalpy become more negative across a period (left to right)?
Across a period, nuclear charge increases and atomic size decreases. Both factors increase the effective attraction between nucleus and incoming electron. Thus, atoms on the right side of the periodic table (non-metals like halogens) release more energy when gaining an electron. For example: ΔegH of halogens is highly negative, while for alkali metals (Na, K) it is low.
Which element has the most negative electron gain enthalpy?
Among all elements, chlorine (Cl) has the most negative electron gain enthalpy (≈ –349 kJ mol⁻¹). This is due to high nuclear charge, optimal atomic size (not too small like F, not too large like Br or I), low electron–electron repulsions compared to fluorine. Hence, chlorine is the most favorable element for gaining an electron.
Why do elements with half-filled or fully filled subshells show irregularities in electron gain enthalpy trends?
Half-filled subshells (e.g., N : 2p³) and fully filled subshells (e.g., Be : 2s², Mg: 3s²) are stable configurations. Adding an extra electron disturbs this stability. Hence, such elements show less negative or even positive electron gain enthalpy values.
Arrange the following in increasing order of electron gain enthalpy : O, F, S, Cl
Electron gain enthalpy values more negative across a period and less negative down a group.
O (–141 kJ/mol), S (–200 kJ/mol), F (–328 kJ/mol), Cl (–349 kJ/mol).
So, order is : O < S < F < Cl.
Between Cl and Br, which has a more negative electron gain enthalpy and why?
Chlorine has a smaller atomic size compared to bromine. Hence, the added electron in chlorine experiences stronger nuclear attraction. Therefore, electron gain enthalpy of Cl is more negative than that of Br.
Predict and explain the sign of electron gain enthalpy of noble gases.
Noble gases have stable octet configurations. Adding an electron requires placing it in a new higher-energy orbital (ns² np⁶ (n+1)s¹), which is energetically unfavourable. Therefore, their electron gain enthalpy is positive.
Why is electron gain enthalpy of oxygen less negative than that of sulfur, even though oxygen is smaller in size?
Oxygen is a second-period element with a very small atomic size. The extra electron must enter a compact 2p orbital, where it faces strong electron–electron repulsions. In sulphur, the incoming electron enters the larger 3p orbital, which is more spacious and experiences less repulsion. Therefore, sulphur releases more energy (ΔegH = –200 kJ/mol) than oxygen (ΔegH = –141 kJ/mol).
Why does phosphorus have a more negative electron gain enthalpy than nitrogen?
The electronic configuration of nitrogen is 1s² 2s² 2p³, i.e., a half-filled stable p³ configuration. Adding one more electron disturbs this stable arrangement, so its ΔegH is almost zero (–7 kJ/mol). While Phosphorus has valence shell electronic configuration is 3s² 3p³. The larger size and less repulsion in 3p orbitals allow easier acceptance of an extra electron. Thus, Phosphorus has a more negative electron gain enthalpy than nitrogen.
Why do second-period elements (N, O, F) have less negative electron gain enthalpy values than their third-period counterparts (P, S, Cl) ?
Second-period elements (N, O, F) are very small. When an additional electron is added, it faces large electron–electron repulsions in compact orbitals. In contrast, third-period elements (P, S, Cl) have larger atomic size and more diffused orbitals, so repulsion is less, and nucleus attraction dominates. Hence, third-period elements have more negative electron gain enthalpies.
Arrange the following elements in order of increasing negativity of electron gain enthalpy : N, P, O, S, F, Cl.
N < P < O < S < F <Cl
N ≈ 0 (stable half-filled p³)
P < O (O smaller, more repulsion)
S > O (larger, less repulsion)
Cl > F (fluorine anomaly due to small size and repulsion).
Why does nitrogen have almost zero electron gain enthalpy?
Nitrogen has the configuration 1s² 2s² 2p³, a half-filled p-orbital (stable configuration). Adding one more electron would disrupt this stability, and the extra electron would face high repulsion in the compact 2p orbital. Hence, nitrogen’s electron gain enthalpy is almost zero.
Why Fluorine has large negative electron gain enthalpy than oxygen ?
Fluorine has large negative electron gain enthalpy than oxygen because Fluorine has a 2s²2p⁵ configuration, needing only one electron to complete its octet its small size and high nuclear charge attract the extra electron strongly.
Important Chapter Interlinks
This section provides a complete and interconnected study of Classification of Elements and Periodicity in Properties, starting with detailed theory and notes for Class 11 Chemistry to build a strong conceptual foundation. You can explore atomic radius and its types including covalent, van der Waals, metallic, and ionic radii to understand periodic trends in atomic size. It also includes Screening Effect (Shielding Effect) : Calculation of Effective or Reduced Nuclear Charge (Slater’s Rules), which explains how inner electrons reduce the nuclear attraction on outer electrons and influence periodic trends. In addition, topics like Radius of Cation is Less and Anion is More Than Its Parent Atom, Size Variation in Isoelectronic Series help explain how ionic size changes due to gain or loss of electrons and how nuclear charge affects size in species with the same number of electrons. The causes of periodicity explain why elements show repeating properties based on electronic configuration, which is further supported by the modern periodic law and structure of the modern periodic table including groups, periods, and blocks for elements even beyond atomic number 100. The historical development is covered through Mendeleev’s periodic law and table, leading to the modern classification of elements into s, p, d, and f blocks with prediction of period, group, and block. To strengthen exam preparation, you can practice JEE Main PYQs, IMU CET PYQs and Merchant Navy sponsorship exam MCQs, and other previous year questions with solutions, along with solved examples, conceptual questions, and practice problems on the modern periodic table. Learn more in this section also to radius of cation is less and anion is more than its parent atom and size variation in Isoelectronic Series. Additionally, complete study material, mock tests, and guidance are provided under Anand Classes Chemistry notes, along with expert support from Er Neeraj Anand, making this section a comprehensive resource for competitive exam preparation. This section also includes detailed study of What is Ionization Enthalpy? Definition, Units, Factors and Successive IE and Ionization Enthalpy Trends Along a Period and Down a Group for better understanding of periodic properties and reactivity of elements.
