Ionization enthalpy trends along a period and down a group explain how the energy required to remove an electron changes across the periodic table. This article covers the general trend across a period, the exceptions at Be-B and N-O, the decrease down a group, and solved NCERT conceptual questions with complete explanations. Understanding these trends is essential for predicting element reactivity and periodic behavior for chemistry exams like CBSE Class 11, JEE, NEET.
How does Ionization Enthalpy Variation with Atomic Number in the Periodic Table ?
Ionization enthalpy provides another example for understanding periodicity among the elements. The ionization enthalpies of some elements are given in Table.1.
| Element | Ionization Enthalpy (kJ mol⁻¹) | Element | Ionization Enthalpy (kJ mol⁻¹) |
| H | 1312 | He | 2372 |
| Li | 520 | Ne | 2080 |
| Be | 899 | Mg | 737 |
| B | 801 | Al | 577 |
| C | 1086 | Si | 786 |
| N | 1402 | P | 1011 |
| O | 1314 | S | 999 |
| F | 1681 | Cl | 1255 |
| Na | 496 | Ar | 1520 |
| K | 419 | Kr | 1350 |
| Rb | 403 | Xe | 1170 |
| Cs | 374 | Rn | 1037 |
The variation of first ionization enthalpy of elements with atomic number (up to atomic number 60) is shown in following Figure.
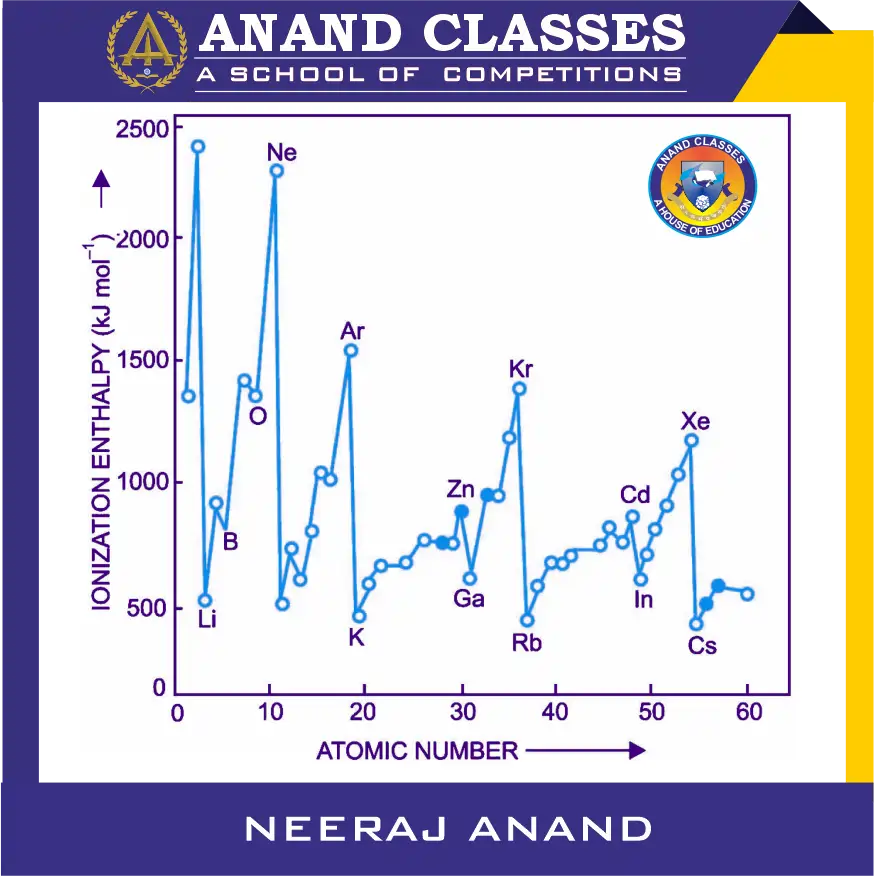
It can be seen that in each period, the maxima are found at the noble gases while minima are found at alkali metals. Thus, the metals of group-I with one electron in outermost s-orbital are easy to ionise while the noble gases (group 18) with ns2 np6 configuration are the most difficult to ionise. The following periodic trends have been observed in the following sections.
“Learn more about Ionization Enthalpy Concept”
Why does Ionization Enthalpy Increase from left to right in a Period ?
In general, the ionization enthalpy increases with increasing atomic number in a period. This is quite clear from the values of ionization enthalpy of the second period elements as given in Figure.
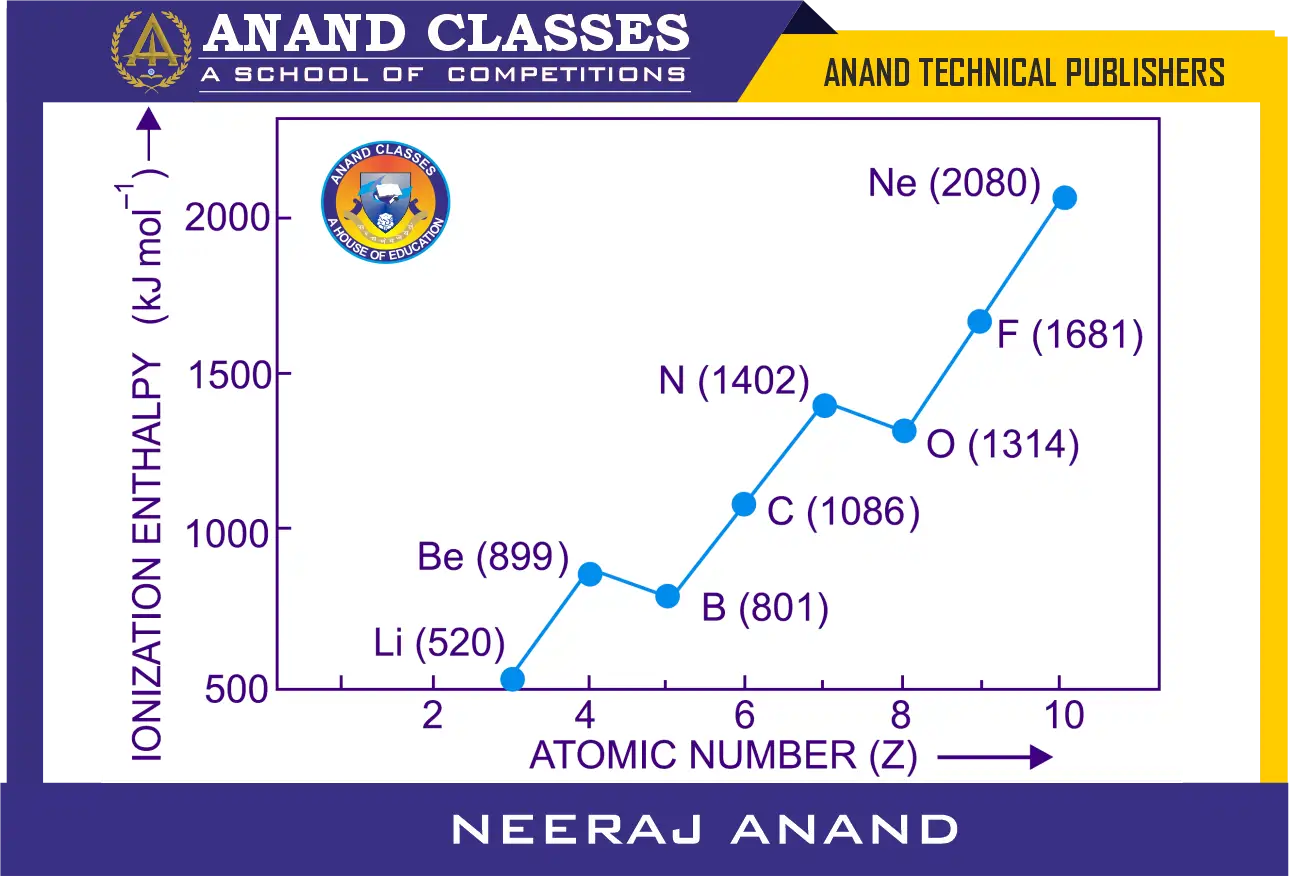
The general increase along a period can be explained on the basis of atomic size and nuclear charge. We know that
(i) on moving across a period from left to right, the nuclear charge increases and there is a greater attraction between the nucleus and the outermost electron.
(ii) the atomic size decreases along a period though the main energy level remains the same so that the valence electrons are closer to the nucleus.
(iii) Shielding effect does not change much, so increased nuclear pull dominates.
As a consequence of increased nuclear charge and simultaneous decrease in atomic size, the valence electrons are more and more tightly held by the nucleus. Therefore, more and more energy is needed to remove the electron and hence, ionization enthalpy keeps on increasing.
“Learn more about atomic size variation“
Explain the Irregularities in the General Trend of Ionization Enthalpy in Second Period of Elements
While the general trend is an increase in Ionization Enthalpy along a period, certain exceptions occur due to half-filled and completely filled subshell stability, and differences in orbital penetration.
Why does ionization enthalpy increase from Lithium (Li) to Beryllium (Be)?
In Li (1s² 2s¹) electron is removed from 2s orbital and in Be (1s² 2s²) electron is removed from a completely filled 2s orbital. Also On moving from Li (Z = 3) to Be (Z = 4), there is an increase in nuclear charge and decrease in atomic size, therefore ionization enthalpy increase from Li to Be.
Why does ionization enthalpy decrease from Beryllium (Be) to Boron (B) despite increase in nuclear charge?
Although the nuclear charge of B is more than Be, yet there is slight decrease in ionization enthalpy from Be to B. This is due to the fact that
(i) the electronic configuration of B (1s2 2s2 2p1) is less stable than that of Be (1s2 2s2) which has completely-filled orbitals.
(ii) When we consider the same principal quantum shell, an s-electron is attracted to the nucleus more than a p-electron. In Be, the electron removed during ionization is a 2s-electron whereas the electron removed during ionization of B is a 2p-electron. Thus we know that the penetration of a 2s-electron to the nucleus is more than that of a 2p-electron and therefore, 2p electron of boron is more shielded from the inner core of electrons than the 2s electron of Be. Hence, it is easier to remove the 2p electron from B compared to the removal of a 2s electron of Be. Thus, B has smaller ionization enthalpy than Be.
As a result the 2p-electron of B is not tightly held by the nucleus as 2s-electron of Be and hence ionization enthalpy of B is less than that of Be.
Why does ionization enthalpy increase from Boron (B) to Carbon (C) to Nitrogen (N) ?
As we move from B to C to N, ionization enthalpy keeps on increasing due to increasing nuclear charge and decreasing atomic size.
Why does nitrogen have relatively high ionization enthalpy that is ionization enthalpy of nitrogen is more than that of oxygen yet nitrogen has less nuclear charge than oxygen?
Oxygen, the element next to nitrogen has slightly smaller ionization enthalpy as compared to that of nitrogen. It is due to the fact that the electronic configuration of nitrogen atom (1s2, 2s2 2px1 2py1 2pz1), in which 2p-orbital is half-filled, is more stable than that of oxygen (1s2, 2s2 2px2 2py1 2pz1). One p-orbital in oxygen contains a paired electron, causing electron–electron repulsion. This repulsion makes it easier to remove one electron from O than from the stable half-filled configuration of N. Therefore, ionization enthalpy of nitrogen is more than that of oxygen.
Why does ionization enthalpy increase from Oxygen (O) to Fluorine (F) to Neon (Ne) and Why Ne have highest ionization enthalpy in a period?
The ionization energy increases from O to F to Ne because of the increased nuclear charge and decrease in size. Neon, the noble gas has the maximum ionization enthalpy in the period due to the stable (ns2 np6) electronic configuration.
Similar variation in ionization enthalpy of the elements of third period has also been observed that is drops observed between Mg to Al and P to S due to the same reasons as Be to B and N to O.
Why does Ionization Enthalpy Decrease from top to bottom in a Group ?
Within a group in the periodic table, there is a gradual decrease in ionization enthalpy as we move from top to bottom. This trend can be clearly observed from the ionization energy values of the elements in the first group as shown in Figure below.
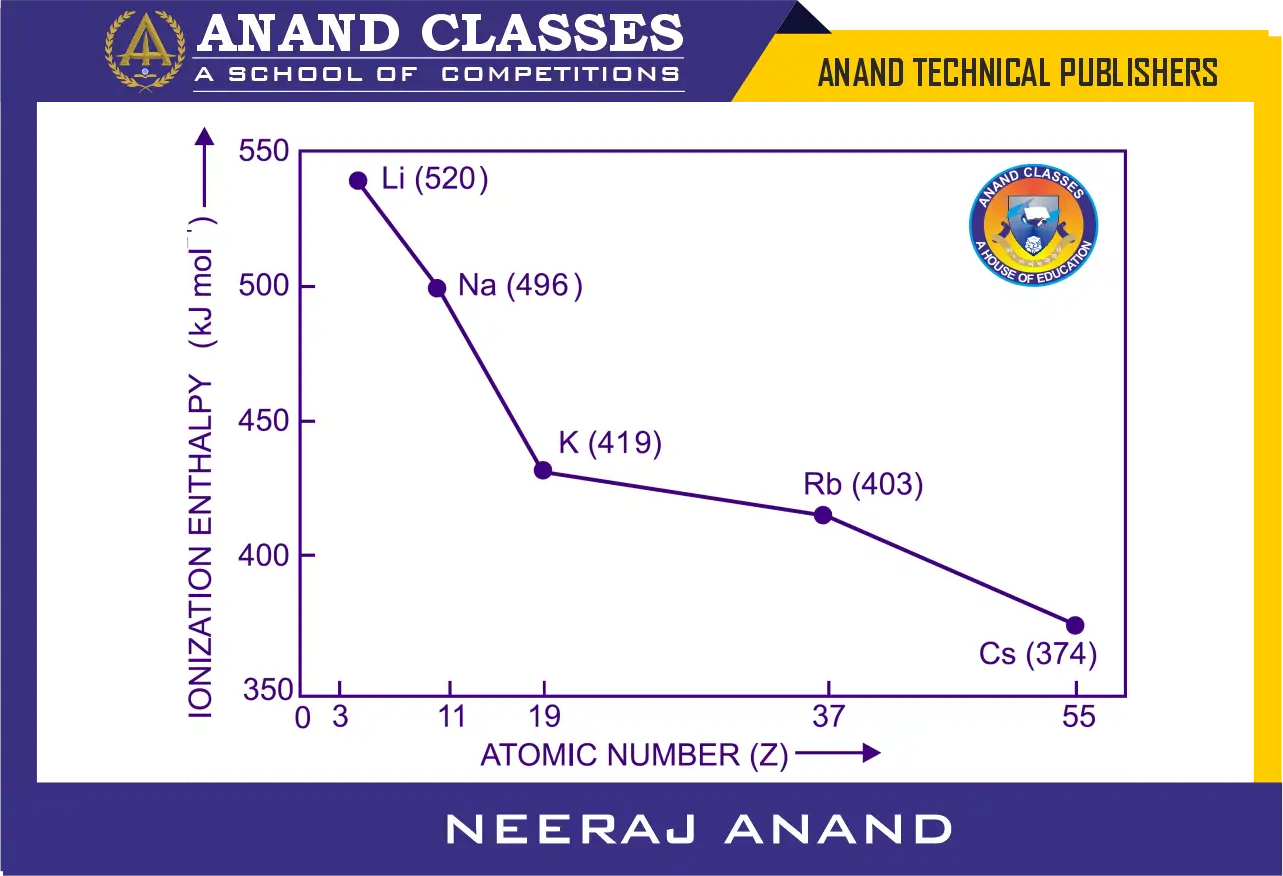
The decrease in ionization enthalpy down a group can be explained by the combined effect of the following factors :
Increase in Nuclear Charge : As we go down a group, each successive element has an extra proton in its nucleus compared to the element above it. This increases the nuclear charge, which tends to pull the electrons closer to the nucleus. On its own, this factor would normally lead to an increase in ionization enthalpy. However, other opposing factors dominate.
Increase in Atomic Size : Moving from top to bottom in a group, each new element has an additional main energy shell (principal quantum number, n). This causes the outermost electron to be located farther from the nucleus. The greater the distance between the nucleus and the outermost electron, the weaker the electrostatic attraction, making it easier to remove the electron and lower the ionization enthalpy.
Increase in Shielding Effect (Screening Effect) : With the addition of inner electron shells as we move down a group, the number of inner electrons increases. These inner electrons repel the outermost electrons and partially block the attractive force of the positively charged nucleus. This shielding effect reduces the effective nuclear charge felt by the outermost electron.
Although the nuclear charge increases down the group, the increase in atomic size and increase in shielding effect are much more significant. These factors overpower the effect of the increased nuclear charge. As a result, the outermost electron becomes less tightly bound to the nucleus. Consequently, less energy is required to remove it. Hence, there is a gradual decrease in the ionization enthalpy in a group.
“Learn more about the factors affecting Ionization Enthalpy (Ionization Energy)”
Ionization Enthalpy Conceptual Questions and Answers for JEE NEET NCERT
Why is the first ionization enthalpy (IE₁) of Li smaller than that of Be, while the second ionization enthalpy (IE₂) of Li is much greater than that of Be? [NCERT]
The difference in the first and second ionization enthalpies of lithium and beryllium can be clearly understood by examining their electronic configurations and the stability of the resulting ions. The observed ionization enthalpy (IE) values are :
| Element | IE₁ (kJ mol⁻¹) | IE₂ (kJ mol⁻¹) |
|---|---|---|
| Li | 520 | 7298 |
| Be | 899 | 1758 |
Electronic configuration explanation in reaction form :
Li ($1s^2 2s^1$) $\xrightarrow{IE_1}$ Li⁺ ($1s^2$) $\xrightarrow{IE_2}$ Li²⁺ ($1s^1$)
Be ($1s^2 2s^2$) $\xrightarrow{IE_1}$ Be⁺ ($1s^2 2s^1$) $\xrightarrow{IE_2}$ Be²⁺ ($1s^2$)
Lithium has the configuration $1s^2 2s^1$, while beryllium has $1s^2 2s^2$. When the first electron is removed, lithium loses its outermost $2s^1$ electron and forms $\text{Li}^+$ with the stable noble gas configuration $1s^2$.
In beryllium, however, the electron is removed from a completely filled $2s^2$ subshell, which is relatively more stable. Because of this extra stability in beryllium, more energy is required to remove the first electron. Therefore, the first ionization enthalpy of lithium is smaller than that of beryllium.
The situation reverses for the second ionization enthalpy. After losing one electron, lithium becomes $\text{Li}^+$ with a stable $1s^2$ configuration. Removing another electron from this stable inner shell requires a very large amount of energy, which explains why the second ionization enthalpy of lithium is extremely high.
On the other hand, $\text{Be}^+$ still has one electron in the $2s$ orbital, and removing this electron does not involve breaking a noble gas configuration. Hence, the second ionization enthalpy of beryllium is much lower than that of lithium.
This explains the overall trend : lithium has a lower first ionization enthalpy but a much higher second ionization enthalpy compared to beryllium.
Learn “Successive ionization enthalpies Concept”
Which of the following pairs of elements would you expect to have lower ionization enthalpy? Explain.
(i) Cl or F (ii) Cl or S (iii) K or Ar (iv) Kr or Xe [NCERT]
(i) In comparing chlorine and fluorine, chlorine has the lower ionization enthalpy. This is because chlorine lies below fluorine in the same group, and as we move down a group, atomic size increases and the outer electron is farther from the nucleus. The increased shielding reduces the nuclear attraction, making it easier to remove an electron.
(ii) Between chlorine and sulfur, sulfur has the lower ionization enthalpy. Both elements lie in the same period, but chlorine is placed to the right of sulfur and has a higher nuclear charge along with a smaller atomic size. As a result, chlorine holds its electrons more tightly, making sulfur easier to ionize.
(iii) In the case of potassium and argon, potassium has a much lower ionization enthalpy. Potassium is an alkali metal with a single loosely bound electron in the $4s$ orbital, whereas argon is a noble gas with a completely filled and highly stable electronic configuration. Removing an electron from argon requires significantly more energy.
(iv) Between krypton and xenon, xenon has the lower ionization enthalpy. As we move down the noble gas group, atomic size increases and shielding becomes more significant, reducing the effective nuclear attraction. This makes electron removal easier in xenon compared to krypton.
From each set, choose the atom which has the largest ionization enthalpy.
(a) F, O, N
(b) Mg, P, Ar
(c) B, Al, Ga
(i) Among nitrogen, oxygen, and fluorine, fluorine has the highest ionization enthalpy. Although nitrogen has a stable half-filled configuration, fluorine has a higher nuclear charge and smallest atomic size (exceptionally small), resulting in a stronger attraction for its outermost electron.
(ii) In the set magnesium, phosphorus, and argon, argon has the largest ionization enthalpy. Being a noble gas with a completely filled shell, argon is extremely stable, and removing an electron requires very high energy.
(iii) For boron, aluminium, and gallium, boron has the highest ionization enthalpy. It is the smallest atom among the three and experiences the least shielding, so its outer electron is held more tightly. Although gallium shows some irregular behavior due to poor shielding by $3d$ electrons, its ionization enthalpy is still lower than that of boron.
How much energy in joules must be needed to convert all the atoms of sodium to sodium ions present in 2.3 mg of sodium vapours? Ionization enthalpy of sodium is 495 kJ/mol (Atomic mass of Na = 23). [NCERT]
The ionization enthalpy of sodium is $495 , \text{kJ mol}^{-1}$, which means 495 kJ of energy is required to ionize one mole of sodium atoms.
Given mass of sodium = $2.3 \text{mg} = 2.3 \times 10^{-3} \text{g}$
No. of moles of sodium present = Given mass/Molar mass = 2.3 × 10−3/23 = 1.0 × 10−4 mol
Therefore, the energy required to ionize 1.0 × 10−4 mol of sodium vapours is : 495 × 10−4 kJ = 0.0495 kJ = 0.0495 × 1000 = 49.5 J
Hence, the energy required to ionize the given amount of sodium is 49.5 J.
The first ionization enthalpy (ΔiH) values of the third period elements, Na, Mg, and Si are respectively 496, 737, and 786 kJ/mol. Predict whether the first ΔiH value for Al will be more close to 575 or 760 kJ/mol. Justify your answer.
The first ionization enthalpy of aluminium is closer to 575 kJ/mol. Although ionization enthalpy generally increases across a period, aluminium shows a deviation from this trend. In aluminium, the electron is removed from a $3p$ orbital, which is higher in energy and less penetrating than the $3s$ orbital of magnesium. As a result, this electron experiences less nuclear attraction and is easier to remove.
Therefore, the ionization enthalpy of aluminium is lower than that of magnesium and is closer to 575 kJ/mol rather than 760 kJ/mol.
Frequently Asked Very Short Type Questions and Answers Based On Ionization Enthalpy – Class 11 JEE NEET
How does ionization enthalpy vary with atomic number in the periodic table?
Ionization enthalpy shows a periodic variation with atomic number. In each period, it generally increases from left to right, reaching a maximum at noble gases, while the lowest values are observed for alkali metals. This pattern reflects the increasing nuclear charge and decreasing atomic size across a period, making electron removal progressively more difficult.
What is the general trend of ionization enthalpy across a period?
Across a period, ionization enthalpy increases from left to right due to increasing nuclear charge and decreasing atomic radius, which results in stronger attraction between the nucleus and valence electrons.
Why does ionization enthalpy increase from left to right in a period?
As we move across a period, the nuclear charge increases while the added electrons enter the same principal energy level. Since shielding does not increase significantly, the effective nuclear attraction on the outermost electrons increases, requiring more energy to remove them.
How does nuclear charge affect ionization enthalpy across a period?
An increase in nuclear charge leads to a stronger attraction between the nucleus and electrons. This increased attraction makes it more difficult to remove an electron, thereby increasing ionization enthalpy.
Why does atomic radius decrease across a period?
Across a period, electrons are added to the same shell while the nuclear charge increases. This pulls the electrons closer to the nucleus, resulting in a decrease in atomic radius.
How does decrease in atomic size influence ionization enthalpy?
When atomic size decreases, the outermost electrons are closer to the nucleus and experience stronger attraction. As a result, more energy is required to remove them, increasing ionization enthalpy.
Why does shielding effect remain nearly constant across a period?
Since electrons are added to the same principal energy level across a period, there is no significant increase in inner-shell electrons. Therefore, shielding effect remains almost constant.
Why are valence electrons more tightly held across a period?
Because of increasing nuclear charge and decreasing atomic size, the attraction between the nucleus and valence electrons increases, making them more tightly bound.
Why is more energy required to remove an electron across a period?
Due to stronger nuclear attraction and reduced atomic size, electrons are held more firmly, so higher energy is needed for their removal.
Why does ionization enthalpy increase from Li to Be?
From Li to Be, nuclear charge increases and atomic size decreases. Additionally, Be has a completely filled 2s subshell, which is more stable, making electron removal more difficult.
Why does ionization enthalpy decrease from Be to B despite increase in nuclear charge?
In boron, the electron is removed from a 2p orbital, which is higher in energy and less penetrating than the 2s orbital of beryllium. Also, Be has a stable fully filled 2s configuration, so it requires more energy to remove an electron than B.
Why are 2p electrons easier to remove than 2s electrons?
2p electrons are less penetrating and more shielded compared to 2s electrons. Therefore, they experience weaker attraction from the nucleus and can be removed more easily.
How does penetration effect explain the lower ionization enthalpy of B?
Since 2s electrons penetrate closer to the nucleus than 2p electrons, they experience stronger attraction. In B, the outer electron is in a 2p orbital, which is less penetrating and more shielded, making it easier to remove.
Why does Be have higher ionization enthalpy than B?
Beryllium has a completely filled 2s subshell which is stable, whereas boron has a partially filled 2p orbital. This makes electron removal from Be more difficult than from B.
Why does ionization enthalpy increase from B to C to N?
As nuclear charge increases and atomic size decreases across these elements, electrons are held more strongly, leading to a steady increase in ionization enthalpy.
Why does nitrogen have relatively high ionization enthalpy?
Nitrogen has a half-filled 2p subshell, which provides extra stability. Removing an electron from this stable configuration requires more energy.
Why does ionization enthalpy decrease from N to O?
In oxygen, one of the p orbitals contains paired electrons, leading to electron–electron repulsion. This makes it easier to remove an electron compared to nitrogen’s stable half-filled configuration.
How does electron–electron repulsion affect ionization enthalpy in oxygen?
The presence of paired electrons in oxygen increases repulsion, reducing the energy required to remove one electron and thus lowering its ionization enthalpy.
Why is half-filled configuration more stable?
Half-filled subshells have symmetrical electron distribution and maximum exchange energy, which increases stability and makes electron removal more difficult.
Why does ionization enthalpy increase from O to F to Ne?
With increasing nuclear charge and decreasing atomic size, electrons are held more tightly. Neon, having a completely filled shell, exhibits maximum stability and thus the highest ionization enthalpy.
Why does neon have the highest ionization enthalpy in a period?
Neon has a completely filled electronic configuration ($ns^2 np^6$), which is highly stable, making electron removal extremely difficult.
What is the trend of ionization enthalpy down a group?
Ionization enthalpy decreases as we move from top to bottom in a group. This happens because atomic size increases and shielding effect becomes stronger with each added inner shell, reducing the effective nuclear attraction on the outermost electrons and making them progressively easier to remove.
How does increase in atomic size affect ionization enthalpy down a group?
As atomic size increases, the outermost electrons are farther from the nucleus, experiencing weaker attraction, which lowers ionization enthalpy.
How does shielding effect change down a group?
Shielding effect increases due to the addition of inner electron shells, which reduces the effective nuclear charge felt by outer electrons.
Why does effective nuclear attraction decrease down a group?
Even though nuclear charge increases, the combined effect of increased distance and shielding reduces the effective attraction on valence electrons.
Why does increase in nuclear charge not increase ionization enthalpy down a group?
The increase in atomic size and shielding effect outweighs the increase in nuclear charge, resulting in a net decrease in ionization enthalpy.
What is the combined effect of atomic size and shielding on ionization enthalpy?
Both increased atomic size and stronger shielding reduce the effective nuclear attraction, making electron removal easier and decreasing ionization enthalpy.
Why are outermost electrons easily removable down a group?
They are farther from the nucleus and are shielded by inner electrons, so they experience weaker attraction and require less energy to remove.
What is the overall conclusion for ionization enthalpy variation down a group?
Ionization enthalpy decreases down a group because increasing atomic size and shielding effect reduce the effective nuclear attraction on the outermost electrons, making them easier to remove.
Why is the first ionization enthalpy (IE₁) of Li smaller than that of Be, while the second ionization enthalpy (IE₂) of Li is much greater than that of Be?
Lithium ($1s^2 2s^1$) loses its outermost $2s$ electron easily, so its IE₁ is low. Beryllium ($1s^2 2s^2$) has a stable filled $2s$ subshell, so more energy is needed to remove an electron, making IE₁ higher. After losing one electron, Li⁺ attains a stable noble gas configuration ($1s^2$), so removing another electron requires very high energy, making IE₂ of Li much greater than that of Be.
Why is the first ionization enthalpy (IE₁) of Li smaller than that of Be?
Lithium ($1s^2 2s^1$) loses its outermost $2s$ electron easily, whereas beryllium ($1s^2 2s^2$) has a stable filled $2s$ subshell. Hence, more energy is required to remove an electron from Be, so IE₁(Be) > IE₁(Li).
Why is the second ionization enthalpy (IE₂) of Li much greater than that of Be?
After losing one electron, Li⁺ attains a stable noble gas configuration ($1s^2$). Removing another electron breaks this stable structure, requiring very high energy. In Be⁺, the second electron is still in the $2s$ orbital, so it is easier to remove. Hence, IE₂(Li) ≫ IE₂(Be).
Which has lower ionization enthalpy : Cl or F?
Cl has lower ionization enthalpy because it has a larger atomic size and greater shielding than F.
Which has lower ionization enthalpy: Cl or S?
S has lower ionization enthalpy because it lies to the left of Cl in the same period and experiences less nuclear attraction.
Which has lower ionization enthalpy: K or Ar?
K has lower ionization enthalpy because it is an alkali metal with a loosely held electron, while Ar is a stable noble gas.
Which has lower ionization enthalpy: Kr or Xe?
Xe has lower ionization enthalpy due to larger atomic size and increased shielding down the group.
Among F, O, and N, which has the largest ionization enthalpy?
F has the largest ionization enthalpy due to highest nuclear charge and smallest size.
Among Mg, P, and Ar, which has the largest ionization enthalpy?
Ar has the largest ionization enthalpy because of its completely filled and stable electronic configuration.
Among B, Al, and Ga, which has the largest ionization enthalpy?
B has the largest ionization enthalpy due to its smaller size and lower shielding effect.
Important Chapter Interlinks
This section provides a complete and interconnected study of Classification of Elements and Periodicity in Properties, starting with detailed theory and notes for Class 11 Chemistry to build a strong conceptual foundation. You can explore atomic radius and its types including covalent, van der Waals, metallic, and ionic radii to understand periodic trends in atomic size. It also includes Screening Effect (Shielding Effect) : Calculation of Effective or Reduced Nuclear Charge (Slater’s Rules), which explains how inner electrons reduce the nuclear attraction on outer electrons and influence periodic trends. In addition, topics like Radius of Cation is Less and Anion is More Than Its Parent Atom, Size Variation in Isoelectronic Series help explain how ionic size changes due to gain or loss of electrons and how nuclear charge affects size in species with the same number of electrons. The causes of periodicity explain why elements show repeating properties based on electronic configuration, which is further supported by the modern periodic law and structure of the modern periodic table including groups, periods, and blocks for elements even beyond atomic number 100. The historical development is covered through Mendeleev’s periodic law and table, leading to the modern classification of elements into s, p, d, and f blocks with prediction of period, group, and block. To strengthen exam preparation, you can practice JEE Main PYQs, IMU CET PYQs and Merchant Navy sponsorship exam MCQs, and other previous year questions with solutions, along with solved examples, conceptual questions, and practice problems on the modern periodic table. Learn more in this section also to radius of cation is less and anion is more than its parent atom and size variation in Isoelectronic Series. Additionally, complete study material, mock tests, and guidance are provided under Anand Classes Chemistry notes, along with expert support from Er Neeraj Anand, making this section a comprehensive resource for competitive exam preparation.
