Atomic radius and its different types—covalent radius, van der Waals radius, metallic radius, and ionic radii—are essential concepts for understanding the size of atoms and ions in chemistry. These radii help explain periodic trends, bonding behavior, and variations in physical and chemical properties of elements across the periodic table. By studying how atomic size changes in different bonding situations, students can better understand concepts like ion formation, lattice structures, and intermolecular interactions.
- What is Atomic Radius ?
- Why Exact Atomic Radius is Difficult to Determine ?
- What is Covalent Radius ?
- Covalent Radius for a homonuclear molecule :
- Covalent Radius for heteronuclear molecules :
- What is Metallic Radius ?
- Covalent Radius versus Metallic Radius
- Atomic Radius – A General Term
- What is Van der Waals Radius ?
- Why Van der Waals Radius is Needed ?
- Why Noble Gases Show Van der Waals Radii ?
- Why Van der Waals Radii is Greater than Covalent Radii ?
- Difference Between Covalent and Van der Waals Radii
- What is Ionic Radius ?
- Measurement of Ionic Radius
- Pauling’s method of determining ionic radius
- Exam Oriented Conceptual Frequently Asked Questions and Answers (FAQs)
What is Atomic Radius ?
The size of an atom is a very important property because many physical and chemical properties are related to it. If the atom is assumed to be spherical, the atomic size is given by the radius of the sphere and is called the atomic radius.
The radius of an atom is the distance between the centre of its nucleus and electrons in the last orbit.
However, according to quantum mechanics, there is no certainty about the exact position of electrons at any time. Theoretically, an electron, at one time, may be very close to the nucleus while at other time it may be far away from the nucleus. In spite of these limitations we need to have some operational definition of the term atomic radius. There are three operational concepts of atomic radius.
- If the bonding is covalent, the radius is called a covalent radius.
- if the two atoms are not bonded by a chemical bond (as in noble gases) the radius is called van der Waal’s radius.
- If the bonding is ionic, the radius is called ionic radius.
Why Exact Atomic Radius is Difficult to Determine ?
Generally, the term atomic radius means the distance from the centre of the nucleus to the outermost shell of electrons. However, it is difficult to determine the exact radius of the atom because of the following reasons :
- Tiny Size of Atoms :
The size of an atom is approximately 1.2 Å or 1.2 × 10⁻¹⁰ m in radius, which is extremely small. - No Well-Defined Boundary :
According to the probability picture of electrons, an atom does not have a well-defined boundary. The probability of finding an electron is never zero—even at large distances from the nucleus. - Atoms Cannot Be Isolated :
It is not possible to isolate an atom and measure its radius directly. The atomic radius is also affected by the presence of other atoms in its neighbourhood. Therefore, the size of an atom may change in going from one environment to another. - Changes in Bonded States :
The size of an atom also changes from one bonded state to another.
Due to the above diificulties, we can therefore defining Atomic Radius :
The distance of closest approach of one atom to another atom in a given bonding situation.
The approximate radii of atoms can be determined by measuring the distance between the centres of two neighbouring atoms (called internuclear distance) in a covalent molecule using :
- X-ray diffraction
- Electron diffraction
- Other spectroscopic techniques
As shown in Figure below, this internuclear distance corresponds to twice the radius of an atom. Therefore, half of this internuclear distance gives the atomic radius.
Atomic radii may be assigned different names depending upon the type of bonding between the atoms such as :
- Covalent radius
- Metallic radius
- van der Waals radius
- Ionic radius
What is Covalent Radius ?
Covalent radius may be defined as one half of the distance between the nuclei of two covalently bonded atoms of the same element in a molecule.
Covalent Radius for a homonuclear molecule :
In the case of molecules containing similar atoms, the covalent radius of an atom may be defined as :
$$r_\text{covalent} = \frac{\text{Internuclear distance between two bonded atoms}}{2}$$
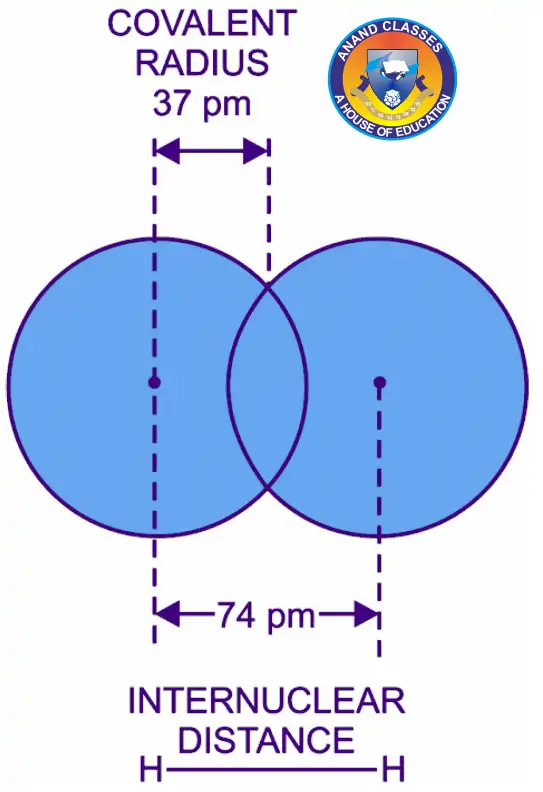
Examples : As shown in Figure above :
- The internuclear distance between two hydrogen atoms in H2 molecule is 74 pm.
- Therefore Atomic radius of hydrogen = 74/2 = 37 pm
- The atomic radii of chlorine and bromine are 99 pm and 114 pm respectively, because the internuclear distances in :
- Cl—Cl = 198 pm
- Br—Br = 228 pm
Covalent Radius for heteronuclear molecules :
In the case of molecules containing different atoms, the covalent radius of an atom may be defined as :
The distance between the centre of the nucleus of the atom and the mean position of the shared pair of electrons between the bonded atoms.
It has been observed that when the covalent radii for different atoms joined by a single covalent bond are added, the resulting value agrees fairly well with the experimentally determined internuclear distance of that molecule.
What is Metallic Radius ?
In the case of metals, a large number of atoms are closely packed and are held together by strong metallic bonds. This close-packed arrangement of metal atoms is known as the metallic lattice.
Metallic radius is defined as one half of the internuclear distance between two neighbouring atoms in a metallic lattice.
Example : In metallic Copper, the distance between two adjacent copper atoms is 256 pm. Therefore Metallic radius of copper = 256/2=128 pm.
Covalent Radius versus Metallic Radius
The covalent radius gives the size of a non-metallic element when atoms are bonded by a single covalent bond in a covalent molecule.
The metallic radius gives the size of a metallic element in a metal where atoms are held together by metallic bonds.
Atomic Radius – A General Term
For simplicity, the term atomic radius may be used for both covalent and metallic radii, depending on whether the element is a non-metal or a metal. However, it must be noted :
Covalent and metallic radii are always shorter than the atomic radii in uncombined atoms.
This is because :
- A covalent bond is formed by the overlapping of atomic orbitals.
- The overlap region becomes common between the two atoms, reducing the distance between their nuclei.
What is Van der Waals Radius ?
Van der Waals radius is defined as half of the internuclear separation of two non-bonded atoms of the same element on their closest possible approach.
or
It is one–half of the distance between the nuclei of two adjacent atoms belonging to two neighbouring molecules of an element in the solid state.
Van der Waals radius is applicable when the atoms are not bonded covalently but are held together by weak Van der Waals forces.

The concept of Van der Waals radius is named after Johannes Diderik van der Waals, who first explained the weak forces of attraction between non-bonded atoms and molecules.
Examples
- Chlorine (Cl2) in solid state :
- Internuclear distance = 360 pm (3.6 Å)
- Van der Waals radius = 360/2 = 180 pm (1.80 Å)
- Hydrogen (H2) in solid state :
- Internuclear distance = 240 pm
- Van der Waals radius = 240/2 = 120 pm (1.2 Å)
Why Van der Waals Radius is Needed ?
- Atomic radius values for most elements are covalent radii because they are measured in covalently bonded molecules.
- Noble gases (e.g., Neon, Argon) do not form covalent molecules like Ne₂ or Ar₂ under normal conditions.
- In their solid state, noble gas atoms are held together by weak van der Waals forces, not by covalent bonds.
- Hence, for noble gases and similar non-bonded atoms, we measure the Van der Waals radius instead of covalent radius.
Why Noble Gases Show Van der Waals Radii ?
- Noble gases (except Xe and Kr) do not form covalent bonds because of their complete octets.
- Their atomic radius cannot be measured as covalent radius.
- Instead, we measure Van der Waals radius, since these atoms exist as discrete monoatomic species and are held together only by Van der Waals forces in the solid state.
- Also known as Inert Gas Radii.
Why Van der Waals Radii is Greater than Covalent Radii ?
The covalent radius is always smaller than the corresponding van der Waal’s radius. This is because of the fact that in the formation of a chemical bond, the two atoms have to come closer to each other. This also explains why covalent bonds are much stronger than the van der Waal’s forces.
It is important to note that since the noble gases ordinarily do not form any covalent bond, in crystals of noble gases, no chemical forces are operating between the atoms. Hence the van der Waal’s forces are the only attractive forces in these cases.
In other words, the van der Waal’s radii constitute the atomic radii of noble gases and since van der Waal’s radii are larger than covalent radii, atomic radii of noble gases are largest in their respective periods (anomaly).
Atoms held by van der Waals forces are farther apart than covalently bonded atoms → van der Waals radii > covalent radii.
Difference Between Covalent and Van der Waals Radii
| Feature | Covalent Radius | Van der Waals Radius |
|---|---|---|
| Nature of Bond | For covalently bonded atoms | For non-bonded atoms held by van der Waals forces |
| Magnitude | Smaller | Larger |
| Measurement State | Molecule in gaseous or solid covalent state | Solid state with weak intermolecular forces |
| Example | H–H bond distance in H2 | Cl–Cl distance in solid Cl2 |
What is Ionic Radius ?
The ionic radius refers to the effective distance from the centre of the nucleus of an ion up to the point where it has an influence in an ionic bond.
Ions are formed when atoms gain or lose electrons :
- Cations : Positively charged ions formed by the loss of electrons (e.g., Na+, Mg2+).
- Anions : Negatively charged ions formed by the gain of electrons (e.g., Cl–, O2-).
Measurement of Ionic Radius
The most common way to measure ionic radius is through X-ray crystallography, which gives the distance between the nuclei of adjacent positive and negative ions in an ionic crystal.
If the ions are treated as spheres, the internuclear distance can be expressed as:
Internuclear Distance = rcation + ranion
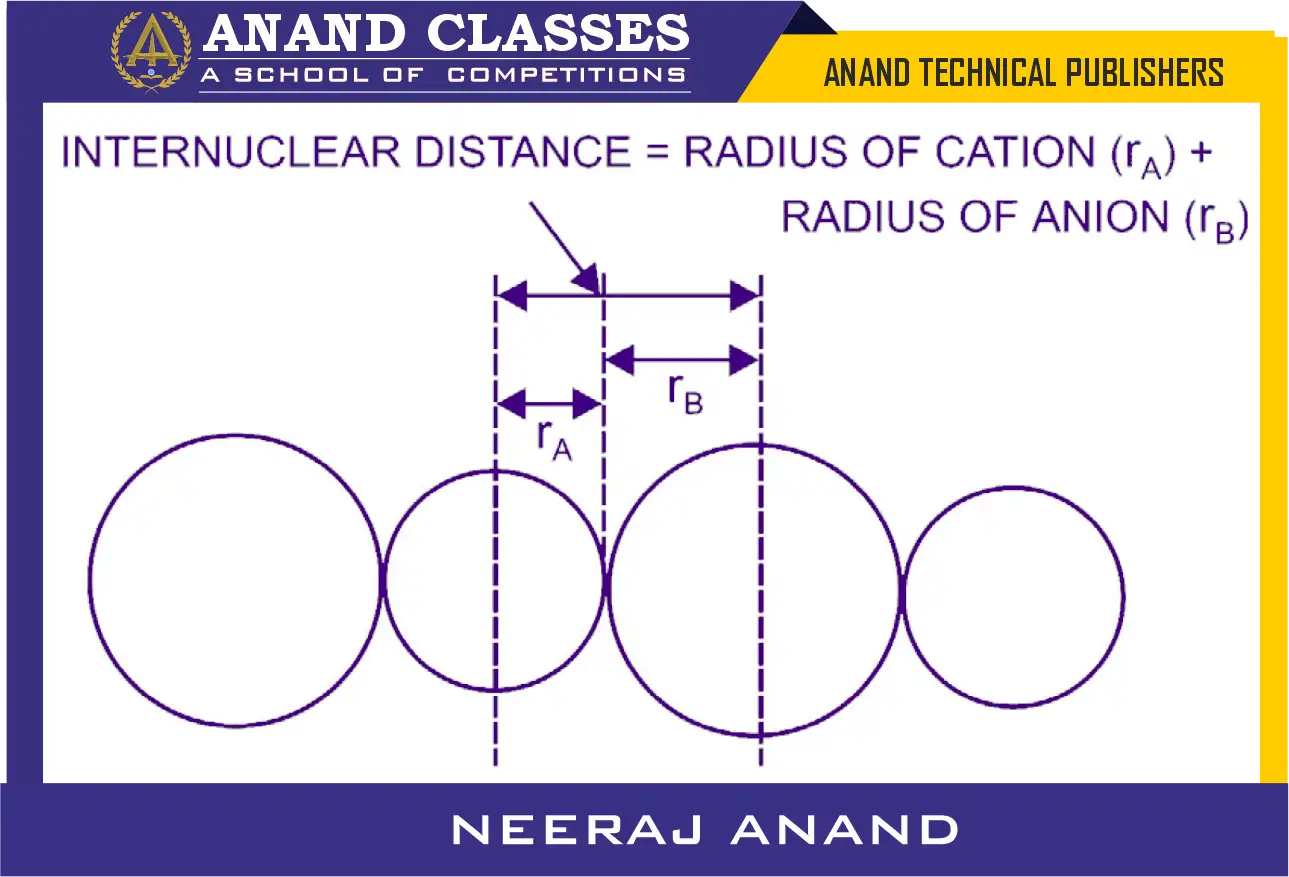
Pauling’s method of determining ionic radius
Pauling’s method is a standard approach that fixes the radius of at least one ion as a reference, allowing calculation of other ionic radii by subtracting from the measured internuclear distances.
Example : In sodium chloride (NaCl), the internuclear distance between Na+ and Cl– is 276 pm. This value is the sum of the ionic radii of Na+ and Cl–. If the radius of one ion is known, the other can be calculated by subtraction.
Exam Oriented Conceptual Frequently Asked Questions and Answers (FAQs)
What is the covalent radius?
The covalent radius is half the distance between the nuclei of two identical atoms joined by a single covalent bond. It is used for non-metals that form covalent compounds.
What is the metallic radius?
The metallic radius is half the distance between the nuclei of two adjacent atoms in a metallic lattice. It applies to metals where atoms are packed in a solid metal structure.
What are the different types of atomic radius?
There are three primary types:
Covalent radius: half the distance between the nuclei of two identical atoms joined by a covalent bond.
Metallic radius: half the distance between nuclei in adjacent atoms within a metallic lattice.
van der Waals radius: half the closest distance between non-bonded atoms in neighboring molecules.
How do covalent and metallic radii differ?
Covalent radius is smaller because of strong directional bonding between atoms.
Metallic radius is larger due to non-directional metallic bonding and looser packing of electron clouds.
Which one is larger for the same element – covalent or metallic radius?
For the same element, the metallic radius is generally larger than the covalent radius due to weaker attraction in metallic bonds compared to covalent bonds.
Give an example comparing covalent and metallic radius.
For sodium (Na):
Covalent radius ≈ 154 pm
Metallic radius ≈ 186 pm
This shows that metallic radius > covalent radius.
Why is the metallic radius applicable only to metals?
Because metallic radius is defined based on the structure of metallic solids, where atoms are arranged in a regular pattern and bonded through a sea of delocalized electrons.
In what types of compounds is covalent radius used?
Covalent radius is used in molecules where atoms are held by covalent bonds, such as in H₂, O₂, N₂, Cl₂, CH₄, etc.
Why are there different measurement methods for atomic radius?
Atoms don’t have clear boundaries, so different contexts—such as bonding type or whether atoms are isolated or in solids—require different definitions and measurement techniques.
Why is the Exact Atomic Radius Difficult to Determine?
The exact atomic radius is difficult to determine because :
Atoms do not have sharp boundaries : The electron cloud around the nucleus does not end abruptly; it gradually fades out, making it hard to define where an atom truly “ends”.
Electron cloud is probabilistic : According to quantum mechanics, electrons exist in orbitals with a probability distribution, not fixed orbits. So, the size of an atom is not precise—it depends on where we consider the edge of the cloud to be.
Atomic radius varies with bonding type : The measured radius changes depending on how the atom is bonded (e.g., covalent, metallic, or van der Waals radius), so there’s no single, fixed value for an atom.
Influenced by surrounding atoms: Atomic size can change based on the chemical environment—whether the atom is isolated, bonded, part of a molecule, or in a crystal lattice.
Relies on indirect measurement : Atomic radius is often calculated using inter-nuclear distances between atoms in molecules or solids, not by measuring individual atoms directly.
Due to the diffuse electron cloud, bonding context, and quantum mechanical nature of electrons, the atomic radius is always an approximate or average value, not an exact measurement.
What is the Van der Waals radius?
The Van der Waals radius is half of the internuclear distance between the nuclei of two adjacent atoms belonging to neighbouring molecules in the solid state. It is measured when atoms are held together by weak Van der Waals forces, not by covalent bonds.
Why is the Van der Waals radius larger than the covalent radius?
Van der Waals forces are weak, so atoms are held at larger distances compared to covalent bonds. This makes the Van der Waals radius always larger than the covalent radius.
Why do noble gases have Van der Waals radii instead of covalent radii?
Noble gases (except Xe and Kr) generally do not form chemical compounds or covalent bonds. Their atomic size is therefore measured using the Van der Waals radius, as in the solid state they are held together only by Van der Waals forces.
Can you give an example of calculating Van der Waals radius?
Yes. The internuclear distance between adjacent chlorine atoms in solid state is 360 pm.
Van der Waals radius = 360/2 = 180 pm or 1.80 Å.
What are some examples of Van der Waals radii for noble gases?
He: 120 pm, Ne: 160 pm, Ar: 191 pm, Kr: 200 pm, Xe: 220 pm
What is ionic radius?
The ionic radius is the effective distance from the nucleus of an ion to the point up to which it exerts an influence in an ionic bond.
How is ionic radius measured?
Ionic radii are generally determined using X-ray diffraction studies, which measure the distance between nuclei of adjacent ions in ionic crystals.
Why is the radius of a cation smaller than its parent atom?
When an atom loses electrons to form a cation, it loses an outer electron shell and experiences increased effective nuclear charge, pulling the remaining electrons closer to the nucleus.
Why is the radius of an anion larger than its parent atom?
When an atom gains electrons to form an anion, electron-electron repulsion increases and the nuclear pull on each electron decreases, resulting in a larger size.
Can ionic radius be the same for cations and anions of the same element?
No. Cations are always smaller, and anions are always larger than their respective parent atoms due to differences in electron configuration and effective nuclear charge.
What is Pauling’s method of determining ionic radius?
Pauling’s method is a standard approach that fixes the radius of at least one ion as a reference, allowing calculation of other ionic radii by subtracting from the measured internuclear distances.
How does ionic radius affect physical properties of compounds?
Ionic radius influences properties like lattice energy, melting point, and solubility — smaller ions generally form stronger ionic bonds and higher melting point compounds.
This section on Classification of Elements and Periodicity in Properties interlinks all important topics starting with the causes of periodicity, explaining why elements show repeating trends based on electronic configuration. It further connects to the historical development through Mendeleev’s periodic table and leads to the modern periodic law and modern periodic table based on atomic number. You will also explore the division of the periodic table into s, p, d, and f blocks, along with the classification of elements as metals, non-metals, and metalloids. To strengthen understanding and exam preparation, practice questions, JEE Main PYQs, and IMUCET PYQs are included, helping you build a complete and connected understanding of periodic trends and properties.
