The historical development of the periodic table marks one of the most important achievements in chemistry. As the number of known elements increased, scientists attempted to classify them based on similarities in their properties. Early efforts by Dobereiner, Newlands, and Lothar Meyer laid the foundation, but it was Dmitri Mendeleev who provided a systematic and successful arrangement of elements. His periodic law and periodic table not only organized the known elements but also predicted the existence and properties of undiscovered ones, making it a major breakthrough in the study of chemistry.
- What is the Need for Classification of Elements?
- Historical Development of The Periodic Table
- 1. Dobereiner’s triads
- 2. De Chancourtois Classification
- 3. Newlands law of octaves
- Mendeleev’s Periodic Law
- Mendeleev’s Periodic Table
- Important Contributions of Mendeleev’s Periodic Table
- Defects of Mendeleev’s Periodic Table
- Very Short Conceptual Question and Answers
- Buy Complete Study Material

What is the Need for Classification of Elements?
Scientists like to find patterns. About 200 years ago, scientists were discovering lots of new elements. However, they were struggling hard to bring order and pattern to the vast amount of information they were discovering about the elements and their properties. The periodic table is an important triumph for the nineteenth century chemists and has become an important landmark in the history of chemistry. It is used as a sorting system for chemical elements. It has become the everyday support for students and teachers and provides a concise organization of the whole of chemistry.
Upto the end of seventeenth century, only 31 elements were known. Therefore, it was very easy to study and remember the properties of these elements. However, during the later part of the eighteenth century, the pace of discovery of new elements quickened. Between 1800 to 1869, the number of identified elements had become nearly double to 63. With such a large number of elements, it became difficult to study individually the chemistry of all these elements and their innumerable compounds. At this stage, it was realised that there should be some simple way to study and remember the numerous properties of the elements and their compounds. This gave rise to necessity of classification of the elements into various groups having similar properties. This has been done by arranging the elements in such a way that similar elements are placed together while dissimilar elements are separated from one another. This is known as classification of elements. Such a classification of the elements has resulted in the formulation of the periodic table.
Periodic table may be defined as the arrangement of the known elements according to their properties in a tabular form.
At present, about 118 elements are known. Of these, the recently discovered elements are man made. Efforts to synthesize new elements are continuing. The periodic classification of the elements has extremely simplified their study. Not only the periodic classification rationalizes the known chemical facts about elements, but it also helps to predict new ones for undertaking further study.
Historical Development of The Periodic Table
Since the beginning of the nineteenth century, scientists have been trying to find a basis of grouping elements having similar properties. Lavoisier classified the elements simply as metals and non-metals. However, this classification proved to be inadequate. Some of the earlier important attempts to classify the elements are briefly summed up below:
1. Dobereiner’s triads
In 1817, a German scientist, Johann Dobereiner classified the elements in groups of three elements called triads.
The elements in a triad had similar properties and the atomic weight of the middle member of each triad is very close to the arithmetic mean (average) of the other two elements.
The common triads of Dobereiner classification were : lithium, sodium and potassium; calcium, strontium and barium; chlorine, bromine and iodine; etc.
Table. Dobereiner’s triads
| Element | Atomic weight | Mean of 1st and 3rd |
|---|---|---|
| Lithium | 7 | |
| Sodium | 23 | $$ \frac{7 + 39}{2} = 23 $$ |
| Potassium | 39 |
| Element | Atomic weight | Mean of 1st and 3rd |
|---|---|---|
| Calcium | 40 | |
| Strontium | 88 | $$ \frac{40 + 137}{2} = 88.5 $$ |
| Barium | 137 |
| Element | Atomic weight | Mean of 1st and 3rd |
|---|---|---|
| Chlorine | 35.5 | |
| Bromine | 80 | $$ \frac{35.5 + 127}{2} = 81.25 $$ |
| Iodine | 127 |
For example, in the triad of lithium, sodium and potassium,
Atomic weight of middle element (sodium) = 23
Mean of atomic mass of lithium and potassium
$$ \frac{7 + 39}{2} = 23. $$
The Dobereiner’s relationship was also referred to as law of triads. However, it seemed to work only for a few elements. It was dismissed as coincidence because all the known elements could not be arranged in triads.
2. De Chancourtois Classification
The next reported attempt was made by a French geologist, A.E.B.de Chancourtois in 1862. He arranged the then known elements in order of increasing atomic weights and proposed a cylindrical table of elements to display the periodic recurrence of properties. He observed that the elements with similar properties fell in a vertical line from the centre of the spiral. However, this did not attract much attention.
3. Newlands law of octaves
In 1865, an English chemist, John Newlands proposed a new system of grouping elements of similar properties.
According to Newlands law of octaves, when the elements are arranged in the increasing order of atomic weights, the properties of every eighth element are similar to the first one.
Newlands called this relation as the law of octaves due to similarity with the musical scale.
This relationship is just like every eighth note resembles the first in octaves of music. The Newlands law of octaves seemed to be true only for elements up to calcium as shown below :
Table. Newlands’s law of octaves
| Element | Li | Be | B | C | N | O | F |
|---|---|---|---|---|---|---|---|
| At. Wt. | 7 | 9 | 11 | 12 | 14 | 16 | 19 |
| Element | Na | Mg | Al | Si | P | S | Cl |
|---|---|---|---|---|---|---|---|
| At. Wt. | 23 | 24 | 27 | 29 | 31 | 32 | 35.5 |
| Element | K | Ca |
|---|---|---|
| At. Wt. | 39 | 40 |
In the above table, sodium (Na), the eighth element from lithium (Li) is similar to Li and similarly the next eighth element potassium is similar to Na. The same is true for magnesium resembling beryllium, aluminium resembling boron, etc.
4. Lother Meyer’s arrangement
In 1869, a German chemist, Lother Meyer used the physical properties such as atomic volume, melting point, boiling point, etc., to arrive at his table of elements. He showed that when the properties of the elements such as atomic volume, melting point, boiling point, etc. are plotted as a function of their atomic weights, the elements with similar properties occupied almost similar positions.
Lothar Meyer proposed that when the elements are arranged in the increasing order of their atomic weights, similarities in physical and chemical properties appear at regular intervals.
All these attempts provided same clue that there are certain regularities among the elements. The first most successful attempt in classification of elements was made by a Russian chemist Dmitri Mendeleev.
Mendeleev’s Periodic Law
In 1869, Mendeleev made a remarkable contribution to the classification of elements. On the basis of physical and chemical properties of the elements, he gave a law known as the periodic law.
Mendeleev’s Periodic Law states that the physical and chemical properties of elements are periodic function of their atomic weights.
This means that when the elements are arranged in the order of their increasing atomic weights, the elements with similar properties repeat at regular intervals. Such orderly recurring properties in a cyclic fashion are said to be occurring periodically. This is responsible for the name periodic law or periodic table.
Mendeleev’s Periodic Table
On the basis of his periodic law, Mendeleev arranged all the known elements in the form of a table known as periodic table. It was observed that the elements with similar properties recur at regular intervals or periodically. As a result of this, the elements fall in certain groups or families. The elements in each group were similar to each other in many properties.
The properties repeated periodically. Mendeleev’s periodic table published in 1905 is given in Table ahead. The horizontal rows in the periodic table are called periods and the vertical columns are called groups.
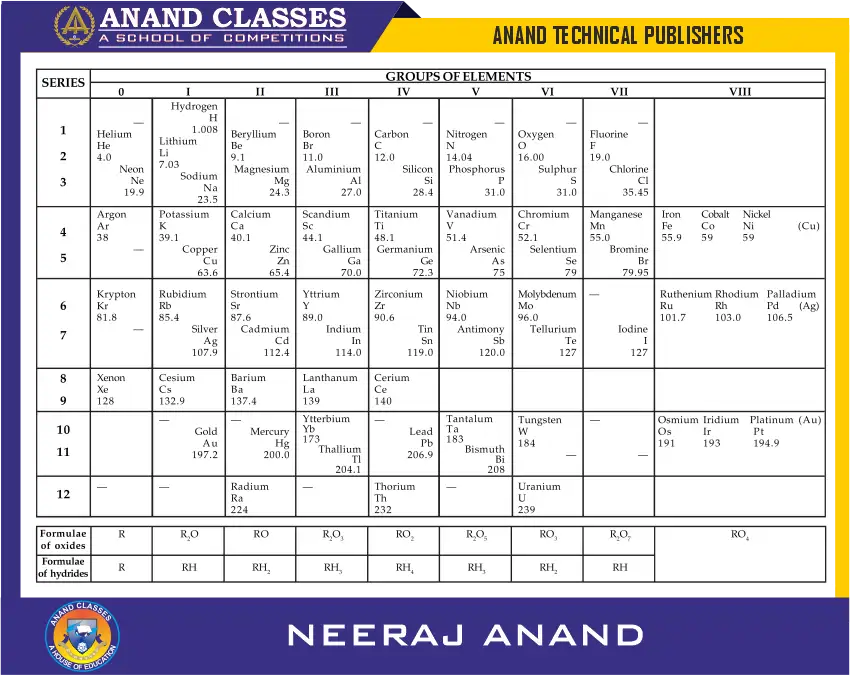
Mendeleev’s system of classifying elements was more elaborate than the earlier attempts. He realized that some of the elements did not fit in very well with his scheme of classification if the order of atomic weight was strictly followed. He showed courage to ignore the order of atomic weights, thinking that the atomic weight measurements might be incorrect. He placed the elements with similar properties together. For example, iodine has lower atomic weight than that of tellurium (of Group VI) but he placed iodine in Group VII alongwith fluorine, chlorine and bromine because of similarities in their properties.
The Mendeleev’s classification gave him so much confidence that he boldly left certain spaces or gaps for undiscovered elements. By considering the properties of the adjacent elements in his table, he predicted the properties of the undiscovered elements. Later on, when these elements were discovered, their properties were found to be exactly similar to those predicted by Mendeleev. For example, gallium and germanium were not discovered at that time, when Mendeleev formulated his periodic table and therefore, he left gaps for these elements. He not only predicted the existence of the elements but he estimated their properties. He tentatively named these elements as eka-aluminium and eka-silicon (from the Sanskrit word eka meaning ‘next’) because he believed that these would be similar to aluminium and silicon respectively. When chemists discovered these elements, Mendeleev’s prediction of their properties proved to be remarkably correct. Some of the properties predicted by Mendeleev for these elements and those found experimentally are given in Table shown below.
Table. Comparison of the properties predicted for eka-aluminium and eka-silicon by Mendeleev with those observed for gallium and germanium.
| Property | Eka-aluminium predicted by Mendeleev | Gallium (found) | Eka-silicon predicted by Mendeleev | Germanium (found) |
|---|---|---|---|---|
| Atomic mass | 68 | 70 | 72 | 72.6 |
| Density (g cm⁻³) | 5.9 | 5.94 | 5.5 | 5.36 |
| Melting point (K) | Low | 29.78 | High | 1231 |
| Formula of oxide | E₂O₃ | Ga₂O₃ | EO₂ | GeO₂ |
| Formula of chloride | ECl₃ | GaCl₃ | ECl₄ | GeCl₄ |
The success of Mendeleev’s bold quantitative predictions convinced Chemists of the usefulness of Mendeleev’s periodic table and led to its wide acceptance.
Important Contributions of Mendeleev’s Periodic Table
Mendeleev’s periodic table was one of the greatest achievements in the development of chemistry. Some of the important contributions of his periodic table are :
- Systematic study of the elements. The Mendeleev’s periodic table simplified the study of chemistry of elements. Knowing the properties of one element in a group, the properties of other elements in the group can be easily guessed. Thus, it became very useful in studying and remembering the properties of a large number of elements.
- Correction of atomic masses. The Mendeleev’s periodic table helped in correcting the atomic masses of some elements based on their positions in the table. For example, atomic mass of beryllium was corrected from 13.5 to 9. Similarly, with the help of this table, atomic masses of indium, gold, platinum etc. were corrected.
- Prediction of new elements. At the time of Mendeleev, only 56 elements were known. While arranging these elements, he left some gaps. These gaps represented the undiscovered elements. Mendeleev predicted the properties of these undiscovered elements on the basis of their positions. For example he predicted the properties of gallium (eka-aluminium) and germanium (eka-silicon) which were discovered later. The observed properties of these elements were found to be similar to those predicted by Mendeleev (Table 4).
Defects of Mendeleev’s Periodic Table
In spite of many advantages, the Mendeleev’s periodic table had certain defects also. Some of these are given below :
- Position of hydrogen. Hydrogen is placed in group I. However, it resembles the elements of group I (alkali metals) as well as the elements of group VIIA (halogens). Therefore, the position of hydrogen in the periodic table is not correctly defined.
- Anomalous pairs. In certain pairs of elements, the increasing order of atomic masses was not obeyed. In these cases, Mendeleev placed elements according to similarities in their properties and not in increasing order of their atomic masses. For example, argon (Ar, atomic mass 39.9) is placed before potassium (K, atomic mass 39.1). Similarly, cobalt (Co, atomic mass 58.9) is placed before nickel (Ni, atomic mass 58.6) and tellurium (Te, atomic mass 127.6) is placed before iodine (I, atomic mass 126.9). These positions were not justified.
- Position of isotopes. Isotopes are the atoms of the same element having different atomic masses but same atomic number. Therefore, according to Mendeleev’s classification, these should be placed at different places depending upon their atomic masses. For example, isotopes of hydrogen with atomic masses 1, 2 and 3 should be placed at three places. However, isotopes have not been given separate places in the periodic table.
- Some similar elements are separated and dissimilar elements are grouped together. In the Mendeleev’s periodic table, some similar elements were placed in different groups while some dissimilar elements had been grouped together. For example, copper and mercury resembled in their properties but they had been placed in different groups. At the same time, elements of group I A such as Li, Na and K were grouped with copper (Cu), silver (Ag) and gold (Au), though their properties are quite different.
- Cause of periodicity. Mendeleev did not explain the cause of periodicity among the elements.
- Position of lanthanoids (or lanthanides) and actinoids (or actinides). The fourteen elements following lanthanum (known as lanthanoids, from atomic number 58–71) and the fourteen elements following actinium (known as actinoids, from atomic number 90—103) have not been given separate places in Mendeleev’s table.
In order to cover more elements, Mendeleev modified his periodic table.
Very Short Conceptual Question and Answers
Q1. What is the need for classification of elements?
The need for classification of elements arose because a large number of elements were discovered, making it difficult to study their properties individually. Classification helps in arranging elements in a systematic manner so that similar elements are grouped together and their properties can be studied easily.
Q2. Why was it easy to study elements earlier?
Earlier, only about 31 elements were known, so it was easy to study and remember their properties individually without any need for classification.
Q3. Why did classification become necessary later?
Between 1800 and 1869, the number of known elements increased to about 63. With such a large number of elements, it became difficult to study their properties individually, which made classification necessary.
Q4. What is meant by classification of elements?
Classification of elements means arranging elements into groups based on similarities in their properties so that similar elements are placed together and dissimilar ones are separated.
Q5. What is a periodic table?
The periodic table is a tabular arrangement of elements based on their properties, where similar elements are grouped together in a systematic manner.
Q6. How does classification help in studying chemistry?
Classification simplifies the study of chemistry by organizing elements into groups, making it easier to understand trends, compare properties, and remember information.
Q7. How many elements are known today?
At present, about 118 elements are known, including some man-made elements.
Q8. What is the main advantage of periodic classification?
The main advantage is that it simplifies the study of elements and helps in predicting the properties of unknown or newly discovered elements.
Q9. What was the first simple classification of elements?
Lavoisier classified elements into metals and non-metals, but this classification was not sufficient.
Q10. What is Dobereiner’s triads?
Dobereiner grouped elements into sets of three called triads, where the atomic mass of the middle element was approximately the average of the other two elements.
Q11. Why was Dobereiner’s classification not successful?
It was not successful because only a few elements could be grouped into triads, and it did not apply to all known elements.
Q12. What was De Chancourtois classification?
He arranged elements in increasing order of atomic weights on a cylindrical spiral, where similar elements appeared in vertical lines.
Q13. What is Newlands law of octaves?
It states that when elements are arranged in increasing order of atomic weights, every eighth element has properties similar to the first element.
Q14. Why did Newlands law fail?
It was valid only up to calcium and did not apply to heavier elements.
Q15. What was Lothar Meyer’s contribution?
He showed that physical properties of elements vary periodically with atomic weight, indicating periodic trends.
Q16. What is Mendeleev’s periodic law?
It states that the physical and chemical properties of elements are periodic functions of their atomic weights.
Q17. What is the significance of Mendeleev’s periodic table?
It arranged elements systematically, predicted new elements, and helped in correcting atomic masses.
Q18. What were the main contributions of Mendeleev’s periodic table?
Systematic study of elements
Prediction of undiscovered elements
Correction of atomic masses
Q19. What are the defects of Mendeleev’s periodic table?
Position of hydrogen is unclear
Anomalous pairs of elements
No proper place for isotopes
Some similar elements separated
Cause of periodicity not explained
Q20. What is the main purpose of classification of elements?
The main purpose is to simplify the study of elements, understand their properties, and predict new elements and their behavior.
Q21. Where Students can read the topic Modern Periodic Law?
Students can read the topic Modern Periodic Law from this link.
Summary
To strengthen your preparation, you can explore Class 11 Chemistry Complete Chapter Notes available on this website. This includes detailed and structured notes for all important chapters such as Some Basic Concepts of Chemistry, Structure of Atom, Classification of Elements and Periodicity in Properties, Chemical Bonding and Molecular Structure, Thermodynamics, Equilibrium, Redox Reactions, Hydrogen, s-Block Elements, p-Block Elements, Organic Chemistry – Some Basic Principles and Techniques, Hydrocarbons, and Environmental Chemistry. Studying these chapters in a step-by-step manner will help you build strong conceptual clarity and improve problem-solving skills for exams like JEE and NEET. For a complete preparation, you can also practice JEE Main Previous Year Questions (PYQs) and revise important concepts through topic-wise notes available on this website. Exploring these connected topics will improve conceptual clarity and help in mastering the entire periodic table effectively.
Buy Complete Study Material
Get comprehensive study material for JEE, NEET, and CBSE Board Class 11 Exams, including detailed notes, MCQs, worksheets, and explanations at Anand Technical Publishers.
Published by: Anand Technical Publishers
Written by: Neeraj Anand
Proprietor: NIRMAL ANAND Educations
Contact: +91-9463138669