The division of the periodic table into s, p, d, and f blocks is based on the electronic configuration of elements and the subshell in which the last electron enters. This classification provides a clear and systematic way to understand the position of elements in the periodic table. By using simple rules, students can easily predict the period, group, and block of any element, which is essential for understanding periodic trends and solving exam-based questions.
- Division of Periodic Table into s, p, d and f block Elements
- What are s-block Elements of the Modern Periodic Table ?
- What are the general characteristics of s-block elements?
- What are p-block Elements of the Modern Periodic Table ?
- What are the general characteristics of p-block elements?
- What are d-block Elements of the Modern Periodic Table ?
- What are the general characteristics of d-block elements?
- What are f-block Elements of the Modern Periodic Table ?
- What are the general characteristics of f-block elements?
- Classification of Elements into four General Types in Modern Periodic Table
- What are Noble gases ?
- What are Representative Elements (s and p-block elements) ?
- What are Transition Elements ?
- What are Inner Transition Elements ?
- Classification of Periodic Table Elements as Metals and Non-Metals
- What are the Advantages of the Long Form of the Periodic Table ?
- What are the Defects of the Long Form of the Periodic Table ?
- Prediction of Period, Group and Block of an Element
- s-Block Elements Rule
- p-Block Elements Rule
- d-Block Elements Rule
- f-Block Elements Rule
- Examples: Finding Period and Group Numbers for Different Block Elements
- Quick Summary Table
- Very Short Conceptual Type Questions and Answers
Division of Periodic Table into s, p, d and f block Elements
The long form of the periodic table can be divided into four main blocks. These are s, p, d and f-blocks. This division of elements is based upon the electronic configurations of the atoms. In this division, the elements which involve the filling of a particular orbital (i.e., s, p, d or f) are grouped together as shown in following Figure. (These are also shown in Long form of the Periodic Table.)
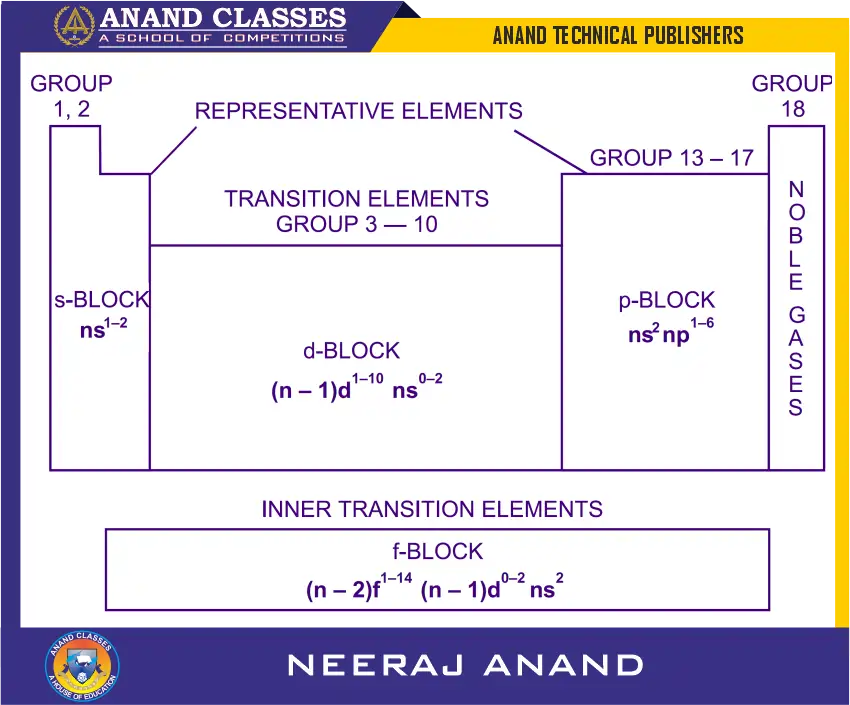
What are s-block Elements of the Modern Periodic Table ?
s-block elements
The elements in which the last electron enters the s-orbital of their outermost energy level are called s-block elements.
s-block elements consists of elements of groups 1 and 2 having the ground state electronic configurations of outermost shell as $ns^1$ and $ns^2$ respectively (where n stands for outermost energy shell). The elements corresponding to $ns^1$ configuration are called alkali metals while those corresponding to $ns^2$ configuration are called alkaline earth metals. Thus, the general electronic configuration of s-block elements may be expressed as:
General electronic configuration of s-block elements : $ns^{1-2}$
What are the general characteristics of s-block elements?
The general characteristics of s-block elements are:
(i) They are soft metals having low melting and boiling points.
(ii) They have low ionisation enthalpies.
(iii) They are very reactive and readily form univalent (alkali metals) or bivalent (alkaline earth metals) ions by losing one or two valence electrons respectively. Because of their high reactivity, they are never found pure in nature.
(iv) They act as strong reducing agents.
(v) Most of them impart characteristic colours to the flame.
(vi) They mostly form ionic compounds except lithium and beryllium.
What are p-block Elements of the Modern Periodic Table ?
p-block elements
The elements in which the last electron enters the p-orbital of their outermost energy level are called p-block elements.
p-block elements of groups 13 to 18 involving addition of one ($ns^2 np^1$), two ($ns^2 np^2$), three ($ns^2 np^3$), four ($ns^2 np^4$), five ($ns^2 np^5$), and six ($ns^2 np^6$) electrons respectively in p-orbitals and s-orbitals are already filled in their atoms constitute p-block. The general electronic configuration for the atoms of this block may be written as:
General electronic configuration of p-block elements : $ns^2 np^{1-6}$
What are the general characteristics of p-block elements?
The general characteristics of p-block elements are:
(i) They include both metals and non-metals. There is a regular gradation from metallic to non-metallic character as we move along a period from left to right in this block. The metallic character increases as we go down the group.
(ii) Their ionisation enthalpies are relatively high as compared to s-block elements.
(iii) They form mostly covalent compounds.
(iv) Some of them show more than one oxidation states in their compounds.
(v) There occurs a gradation from reducing to oxidising properties as we move across the periods of this block.
| What are representative elements ? |
|---|
| The elements of s- and p-block are collectively called representative elements. |
The elements of last group (18) having $ns^2 np^6$ configuration are called noble gases. All the orbitals in the valence shell of the noble gases are completely filled and they have no tendency to lose or gain electrons. Therefore, the noble gases exhibit very low reactivity. Preceding the noble gas family are two chemically important groups of non-metals. These are halogens (group 17) and chalcogens (group 16). These two groups of elements can readily accept one and two electrons respectively to attain noble gas configurations and form univalent and divalent negative ions. In these elements, the non-metallic character increases as we move from left to right across a period and metallic character increases as we go down the group.
What are d-block Elements of the Modern Periodic Table ?
d-block elements
The elements in which the last electron enters the d-orbitals of their last but one (called penultimate) energy level constitute d-block elements.
This block consists of the elements lying between s and p blocks starting from fourth period and onwards. They constitute groups 3 to 12 in the periodic table. In these elements the outermost shell contains one or two electrons in their s-orbital ($ns$) but the last electron enters the last but one d-subshell i.e., $(n-1)d$. The general electronic configuration for the atoms of d-block may be written as:
General electronic configuration : $(n-1)d^{1-10} ns^{0-2}$
Thus, it is the d-orbital which is progressively being filled in the elements of this block.
| Why d-block elements are called transition elements ? |
|---|
| d-block elements are called transition elements because the properties of these elements are midway between those of s-block and p-block elements. In a way, transition metals form a bridge between the chemically active metals of s-block elements and the less active elements of Groups 13 and 14. Therefore, they represent transition (change) in behaviour and take their familiar name “transition elements.” |
The d-block comprises three complete rows of ten elements and one incomplete row. These rows are called first, second and third transition series which involve the filling of 3d, 4d and 5d-orbitals respectively. These series are also called transition series.
First transition series
Scandium (Z = 21) to Zinc (Z = 30)
(4th period : 3d-orbitals are gradually filled)
Second transition series
Yttrium (Z = 39) to Cadmium (Z = 48)
(5th period : 4d-orbitals are gradually filled)
Third transition series
Lanthanum (Z = 57), Hafnium (Z = 72) to Mercury (Z = 80)
(6th period : 5d-orbitals are gradually filled)
Fourth transition series
Actinium (Z = 89), Rutherfordium (Z = 104) to Copernicium (Z = 112)
(7th period : 6d-orbitals are gradually filled)
What are the general characteristics of d-block elements?
The general characteristics of d-block elements are:
(i) They are metals having high melting and boiling points.
(ii) Most of them form coloured compounds.
(iii) They have a good tendency to form complex compounds.
(iv) Their compounds are generally paramagnetic.
(v) They exhibit several oxidation states.
(vi) Most of the transition elements such as Mn, Ni, Co, Cr, V, Pt and their compounds are used as catalysts.
What are f-block Elements of the Modern Periodic Table ?
f-block elements
The elements in which the last electron enters the f-orbitals of their atoms are called f-block elements.
In f-block elements, the last electron is added to the third to the outermost (called antipenultimate) energy level; $(n-2)f$. These consist of two series of elements placed at the bottom of the periodic table:
(i) The first series follows lanthanum, La (Z=57) and the elements present in this series (58Ce – 71Lu) are called lanthanoids or lanthanides. These are also called rare earth elements.
(ii) The second series follows actinium, Ac (Z = 89) and the elements present in this series (90Th – 103Lr) are called actinoids or actinides. These are of radioactive nature.
The general electronic configuration of f-block elements may be written as:
General electronic configuration : $(n-2)f^{1-14} (n-1)d^{0-2} ns^2$
For example, for lanthanoids, the general electronic configuration is $4f^{1-14} 5d^{0-1} 6s^2$ while for actinoids, it is $5f^{1-14} 6d^{0-2} 7s^2$.
The elements included in these two series are called inner transition elements, because they form transition series within the transition elements of d-block.
What are the general characteristics of f-block elements?
The general characteristics of f-block elements are:
(i) They are heavy metals.
(ii) They generally have high melting and boiling points.
(iii) They exhibit variable oxidation states.
(iv) They form coloured ions.
(v) They have the tendency to form complex compounds.
(vi) Actinoids are radioactive in nature. Many of the actinoid elements have been made only in nanogram quantities or even less by nuclear reactions and their chemistry is not fully studied. The elements after uranium are called transuranium elements.
Classification of Elements into four General Types in Modern Periodic Table
On the basis of above four blocks, the elements can be classified into the following four general types :
What are Noble gases ?
The noble gases are found at the end of each period in group 18. Except for helium, the elements have completely filled s- and p-orbitals of the outermost shell i.e., $ns^2 np^6$. Helium has $1s^2$ configuration. All these elements are highly stable and chemically inert under ordinary conditions.
What are Representative Elements (s and p-block elements) ?
All the elements of s and p-block with exception of noble gases are called representative elements. They represent two groups 1 (alkali metals) and 2 (alkaline earth metals) on the extreme left and five groups from group 13 to 17 on the right hand side of the periodic table.
What are Transition Elements ?
The elements of d-block elements are called transition elements. These include elements of group 3 to 12 lying in between the representative elements (between s and p-blocks).
What are Inner Transition Elements ?
The elements of f-block are called inner transition elements. These comprise two series of 14 elements called lanthanoids and actinoids.
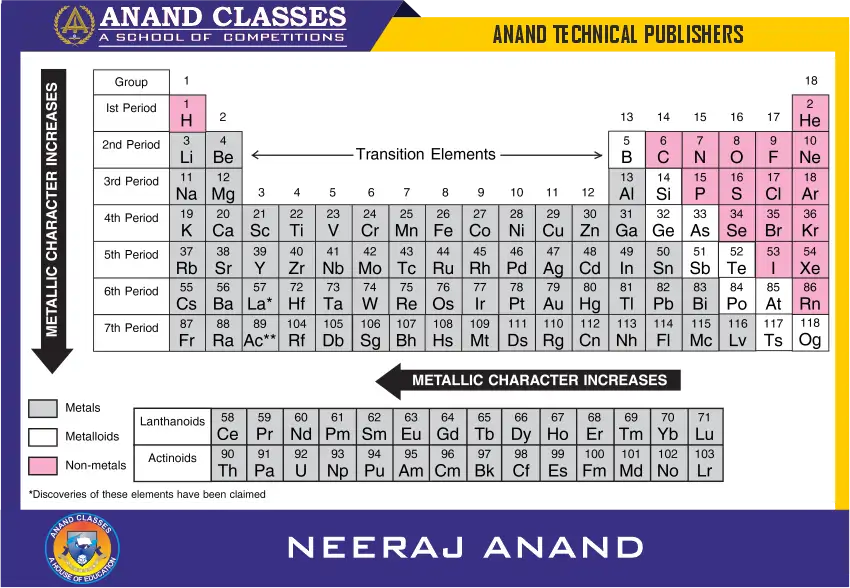
Classification of Periodic Table Elements as Metals and Non-Metals
In addition to classifying the elements into s-, p-, d- and f–block, the elements can be classified on the basis of their properties as:
(i) Metals and
(ii) Non-metals
Metals comprise more than 78% of all known elements and appear on the left hand side of the Periodic Table. They have high melting and boiling points and are generally solid at room temperature (mercury is an exception which is a liquid at room temperature). Gallium and caesium also have very low melting points, 303 K and 302 K respectively. They are good conductors of heat and electricity. They are malleable (can be flattened into thin sheets) and ductile (can be drawn into wires). They have bright lustre.
On the other hand, non-metals are present on the right hand side of the periodic table. They are usually solids or gases at room temperature with low melting and boiling points (boron and carbon are exceptions). The only liquid non-metal is bromine. Most non-metals are brittle and are neither malleable nor ductile.
In general, the elements become more metallic as we go down a group and non-metallic character increases as we move from left to right across a period in the Periodic Table. It may be noted that the change from metallic to non-metallic character is not abrupt as shown by zig-zag line in Figure. The elements bordering this line (silicon, germanium, arsenic, antimony, tellurium, etc.) show properties characteristic of both metals and non-metals. These are called metalloids or semimetals.
What are the Advantages of the Long Form of the Periodic Table ?
The important advantages of the long form of the periodic table are given below:
- This classification is based on the atomic number which is a more fundamental property of the elements.
- Since this classification is based on the atomic number and not on the atomic mass, the position of placing isotopes at one place is fully justified.
- The position of elements in the periodic table is governed by the electronic configurations, which determine their properties.
- It is easy to remember and reproduce.
- The systematic grouping of elements into four blocks; s, p, d and f has made the study of the elements more simple.
- The position of some elements which were misfit on the basis of atomic mass is now justified on the basis of atomic number. For example, argon proceeds potassium because argon has atomic number 18 and potassium has 19.
- The lanthanoids and actinoids which have properties different from other groups are placed separately at the bottom of the periodic table.
What are the Defects of the Long Form of the Periodic Table ?
Although the Long Form of the periodic table has helped in systemising the study of chemistry of elements, yet it has certain defects. The main defects of this table are:
- The position of hydrogen is not settled. It resembles with alkali metals as well as halogens.
- Lanthanoids and actinoids have not been accommodated in the main body of the periodic table.
Prediction of Period, Group and Block of an Element
The period, group or block of an element can be predicted from the electronic configuration of the elements.
- Period of the element corresponds to the principal quantum number (n) of the valence shell.
- Block of the element corresponds to the subshell (s,p,d,f) which receives the last electron.
- Group is predicted from the number of electrons in the outermost shell or penultimate (last but one, i.e., (n-1) shell as follows :
s-Block Elements Rule
General Electronic Configuration for s-block elements is ns1−2.
Period number of s-block elements is the value of n (principal quantum number) of the outermost shell.
Group number of s-block element is equal to number of valence electrons ($ns$ electrons) in the outermost shell.
- If ns1 → Group 1 (Alkali metals)
- If ns2 → Group 2 (Alkaline earth metals)
Example 1: Sodium (Na), Z = 11
Configuration: 1s² 2s² 2p⁶ 3s¹
Outer shell: n = 3 → Period 3
ns¹ → Group 1
Example 2: Calcium (Ca), Z = 20
Configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s²
Outer shell: n = 4 → Period 4
ns² → Group 2
p-Block Elements Rule
General Electronic Configuration for p-block elements is ns2np1−6.
Period number of p-block elements is the value of n (principal quantum number) of the outermost shell.
Group number of p-block element is equal to 10 + number of valence electrons ($ns$ and $np$) in the outermost shell.
Example 1: Nitrogen (N), Z = 7
Configuration: 1s² 2s² 2p³
Outer shell: n = 2 → Period 2
Group = 10 + (2 + 3) = 15
Example 2: Chlorine (Cl), Z = 17
Configuration: 1s² 2s² 2p⁶ 3s² 3p⁵
Outer shell: n = 3 → Period 3
Group = 10 + (2 + 5) = 17
Example 3: Oxygen (O), Z = 8
Configuration: 1s² 2s² 2p⁴
Outer shell: n = 2 → Period 2
Group = 10 + (2 + 4) = 16
For d-block element, group number is equal to total number of electrons in $(n-1)d$ and $ns$ subshells.
d-Block Elements Rule
General Electronic Configuration for d-block elements is (n−1)d1−10ns0−2.
Period number of d-block elements is the value of n (principal quantum number) in the outermost ns orbital.
Group number of d-block element is equal to (electrons in (n–1)d) + (electrons in ns).
Example 1: Iron (Fe), Z = 26
Configuration: [Ar] 3d⁶ 4s²
n = 4 → Period 4
Group = 6 (d-electrons) + 2 (s-electrons) = Group 8
Example 2: Copper (Cu), Z = 29
Configuration: [Ar] 3d¹⁰ 4s¹
n = 4 → Period 4
Group = 10 + 1 = Group 11
Example 3: Chromium (Cr), Z = 24 (exception)
Configuration: [Ar] 3d⁵ 4s¹
n = 4 → Period 4
Group = 5 + 1 = Group 6
f-Block Elements Rule
General Electronic Configuration for d-block elements is (n−2)f1−14(n−1)d0−2ns2.
Period number of f-block elements is the value of n (principal quantum number) in the outermost ns orbital.
- Lanthanides → n = 6 → Period 6
- Actinides → n = 7 → Period 7
All f-block elements are placed in Group 3.
Example 1: Cerium (Ce), Z = 58
Configuration: [Xe] 4f¹ 5d¹ 6s²
n = 6 → Period 6
Group = 3
Example 2: Uranium (U), Z = 92
Configuration: [Rn] 5f³ 6d¹ 7s²
n = 7 → Period 7
Group = 3
Examples: Finding Period and Group Numbers for Different Block Elements
| Block | Element (Symbol, Z) | Electronic Configuration | Step to Find Period | Step to Find Group | Result |
|---|---|---|---|---|---|
| s-block | Sodium (Na, 11) | 1s² 2s² 2p⁶ 3s¹ | Outermost n = 3 → Period 3 | ns¹ → Group 1 | Period 3, Group 1 |
| s-block | Calcium (Ca, 20) | 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² | Outermost n = 4 → Period 4 | ns² → Group 2 | Period 4, Group 2 |
| p-block | Oxygen (O, 8) | 1s² 2s² 2p⁴ | Outermost n = 2 → Period 2 | Group = 10 + (2+4) = 16 | Period 2, Group 16 |
| p-block | Chlorine (Cl, 17) | 1s² 2s² 2p⁶ 3s² 3p⁵ | Outermost n = 3 → Period 3 | Group = 10 + (2+5) = 17 | Period 3, Group 17 |
| d-block | Iron (Fe, 26) | [Ar] 3d⁶ 4s² | Outermost n = 4 → Period 4 | Group = 6 + 2 = 8 | Period 4, Group 8 |
| d-block | Copper (Cu, 29) | [Ar] 3d¹⁰ 4s¹ | Outermost n = 4 → Period 4 | Group = 10 + 1 = 11 | Period 4, Group 11 |
| d-block | Chromium (Cr, 24) | [Ar] 3d⁵ 4s¹ | Outermost n = 4 → Period 4 | Group = 5 + 1 = 6 | Period 4, Group 6 |
| f-block | Cerium (Ce, 58) | [Xe] 4f¹ 5d¹ 6s² | Outermost n = 6 → Period 6 | All f-block = Group 3 | Period 6, Group 3 |
| f-block | Uranium (U, 92) | [Rn] 5f³ 6d¹ 7s² | Outermost n = 7 → Period 7 | All f-block = Group 3 | Period 7, Group 3 |
| Tips for Students |
|---|
Always write the full electron configuration first. Identify the highest value of n → gives the period number. Apply the specific group formula based on the block. Remember exceptions in d-block (Cr, Cu, Mo, Ag, etc.) where electrons shift for stability. |
Quick Summary Table
| Block | General Configuration | Period Rule | Group Rule |
|---|---|---|---|
| s-block | ns¹–² | n | ns¹ → 1, ns² → 2 |
| p-block | ns² np¹–⁶ | n | 10 + (s + p electrons) |
| d-block | (n–1)d¹–¹⁰ ns⁰–² | n | (d electrons + s electrons) |
| f-block | (n–2)f¹–¹⁴ (n–1)d⁰–² ns² | n | Always Group 3 |
Very Short Conceptual Type Questions and Answers
What are s-Block elements?
S-Block elements are those whose outermost electrons occupy an s-orbital, comprising Group 1 (alkali metals) and Group 2 (alkaline earth metals). Some sources also consider hydrogen and helium—though helium is a noble gas, it has its electron in the s-orbital.
How many s-Block elements are there, and which are they?
There are 14 elements:
Group 1: Hydrogen (H), Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Caesium (Cs), Francium (Fr)
Group 2: Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba), Radium (Ra).
What is the general electronic configuration of s-block elements ?
Group 1: Outer electron in ns¹
Group 2: Two outer electrons in ns²
In both groups, the valence electron is in an s‑orbital, which defines their chemistry.
What are the general physical properties of s-block elements ?
Soft, silvery-white metals
Low melting and boiling points
Low densities, especially in Group 1
Good conductors of heat and electricity
These trends arise due to their larger size, electropositive nature, and simple electronic structure
What are the general chemical properties of s-block elements ?
Highly reactive, especially with water and oxygen
Electropositive with low ionization energies, making them strong reducing agents
Form basic oxides and hydroxides, like NaOH, Ca(OH)₂
Reactivity increases down the group.
What is the “diagonal relationship” in s-Block?
Certain elements show similarities across diagonal positions in adjacent groups:
Li and Mg
Be and Al
These pairs behave similarly due to comparable ionic sizes and polarizing power.
Why are s-Block elements called strong reducing agents?
Because of their low ionization energy, they easily lose electrons. As a result, they reduce other species by donating electrons, and themselves oxidize easily.
What flame colors do s-Block elements give and why is that significant?
When heated in flame tests:
Sodium gives a bright yellow
Potassium gives lilac/purple
Calcium gives brick-red
These distinct colors are due to electronic transitions and are used for qualitative tests.
Where s-Block elements appear in everyday life & industry?
Sodium (as NaCl) in food, water treatment, chemical manufacturing
Calcium in bones, cement, and plaster
Magnesium and other metals used in fireworks, fertilizers, antacids, and batteries
They’re also vital for metallurgy and chemical synthesis.
Are there hazards associated with handling s-Block elements?
Yes. Because many are highly reactive:
Alkali metals (e.g. Na, K, Cs) can ignite with moisture or oxygen
Must be stored under mineral oil or inert gas and handled with proper safety precautions
Some elements (e.g. Be, Ra) may be toxic or radioactive.
What are p-Block elements?
p-Block elements are those in which the last electron enters the p-orbital of the valence shell. They are placed in Groups 13 to 18 of the modern periodic table.
What is the general electronic configuration of p-Block elements?
The general electronic configuration is: ns2 np1−6
This means they have 2 electrons in the s-subshell and 1 to 6 electrons in the p-subshell of the outermost shell.
Which groups of the periodic table belong to the p-Block?
The p-Block includes:
Group 13 → Boron family
Group 14 → Carbon family
Group 15 → Nitrogen family (Pnictogens)
Group 16 → Oxygen family (Chalcogens)
Group 17 → Halogens
Group 18 → Noble gases
What types of elements are found in the p-Block?
The p-Block is the most diverse:
Metals → e.g., Al, Sn, Pb
Non-metals → e.g., C, N, O, F
Metalloids → e.g., B, Si, As
Why are p-Block elements chemically diverse?
Because they have variable valence shell electrons (np¹ → np⁶), leading to different oxidation states and bonding types. The block contains everything from highly reactive halogens to inert noble gases.
What are common oxidation states in p-Block?
Group 13 → +3, +1
Group 14 → +4, +2
Group 15 → –3, +3, +5
Group 16 → –2, +4, +6
Group 17 → –1, +1, +3, +5, +7
Group 18 → 0 (inert), but Xe shows +2, +4, +6, +8
Which essential life elements belong to the p-Block?
Carbon (C) → basis of organic compounds
Nitrogen (N) → proteins, DNA
Oxygen (O) → respiration
Phosphorus (P) → ATP, DNA
Sulphur (S) → amino acids
Which group in p-Block is most reactive?
Halogens (Group 17) are the most reactive non-metals due to their high electronegativity and strong tendency to gain one electron to complete their octet.
Why are noble gases chemically inert?
Because they have a completely filled valence shell (ns² np⁶), making them stable and unreactive.
Why do heavier p-Block elements show lower oxidation states?
Due to the inert pair effect — the reluctance of the ns² electrons to participate in bonding as atomic size increases.
Why are d-Block elements called transition elements?
They are called transition elements because their properties are intermediate (or transitional) between highly reactive s-block metals and less reactive p-block elements.
Which transition metal is liquid at room temperature?
Mercury (Hg) is the only transition metal that exists as a liquid at room temperature.
Why do most transition metals form coloured compounds?
Because of d–d electronic transitions within partially filled d-orbitals.
Why are Zn, Cd, and Hg not considered true transition metals?
Their stable ions (Zn2+, Cd2+, Hg2+) have completely filled d-orbitals (d10 configuration), so they do not show variable oxidation states or coloured ions.
Why are transition metals good catalysts?
Due to their ability to change oxidation states and form intermediate complexes, which lower the activation energy of reactions.
What are f-Block elements?
f-Block elements, also called inner transition elements, are those in which the (n–2)f orbitals (inner-penultimate shell) are progressively filled. They are placed separately at the bottom of the periodic table and consist of two series — lanthanides (4f) and actinides (5f).
Why are they called inner transition elements?
They are called inner transition elements because the electrons are added to an inner f-subshell (third shell from the outermost) while the outer shells remain similar in configuration.
What is the general electronic configuration of f-Block elements?
General configuration: [Noble gas] (n–2)f1-14 (n–1)d0-2 ns2
Example: Ce → [Xe] 4f¹ 5d¹ 6s².
What is the difference between lanthanides and actinides?
Lanthanides (4f): Mostly +3 oxidation state, generally stable, mostly non-radioactive.
Actinides (5f): Show multiple oxidation states (+3 to +7), all radioactive, many synthetic (transuranic).
Why do many f-Block elements form coloured ions?
Because of f–f electronic transitions within partially filled f-orbitals, which absorb certain wavelengths of light, producing colour.
Why are most actinides radioactive?
Actinide nuclei are large and unstable due to poor shielding by 5f electrons, which leads to radioactive decay such as α-decay, β-decay, or spontaneous fission.
What is lanthanide contraction?
It is the gradual decrease in ionic and atomic radii from Ce³⁺ to Lu³⁺ due to poor shielding of nuclear charge by 4f electrons.
Give some uses of f-Block elements.
Lanthanides: Neodymium magnets, glass polishing (CeO₂), lighter flints (mischmetal).
Actinides: Uranium & Plutonium in nuclear reactors, Americium in smoke detectors, Thorium in gas mantles.
What are s-Block elements?
s-Block elements are those in which the valence electrons enter the s-orbital. They include Group 1 (alkali metals) and Group 2 (alkaline earth metals). They are highly reactive metals with low ionization energies and form ionic compounds.
Which groups belong to the p-Block?
The p-Block consists of Groups 13 to 18 of the periodic table. The valence electrons enter the p-orbital and the block contains metals, non-metals, and metalloids.
Why are d-Block elements called transition elements?
d-Block elements have partially filled d-orbitals in their atoms or ions and form a transition between s- and p-blocks in the periodic table. They show properties like variable oxidation states, coloured ions, and catalytic activity.
Why are f-Block elements called inner transition elements?
Because their (n–2)f orbitals (inner-penultimate shell) are being filled. They are placed separately at the bottom of the periodic table and include lanthanides (4f) and actinides (5f).
Which block contains only metals?
The s-block, d-block, and f-block contain only metals, whereas the p-block contains metals, non-metals, and metalloids.
Which block contains radioactive elements?
Some d-block elements (e.g., Tc) are radioactive.
All actinides (f-block) are radioactive.
Which block shows the most variable oxidation states?
The d-block elements (transition metals) and actinides (f-block) show the most variable oxidation states due to close energy levels of orbitals.
What is lanthanide contraction?
A gradual decrease in ionic and atomic radii across the lanthanide series (Ce³⁺ to Lu³⁺) due to poor shielding of nuclear charge by 4f electrons.
Where are metals located in the periodic table?
Metals are located on the left and center of the periodic table, covering s-block, d-block, lower p-block, and all of the f-block.
Where are non-metals located in the periodic table?
Non-metals are located on the right side of the periodic table, mainly in the upper p-block (Groups 14–18), plus hydrogen in Group 1.
What is the zig-zag line in the periodic table?
The zig-zag (stair-step) line separates metals on the left from non-metals on the right. Elements along this line are metalloids.
Which elements are metalloids?
Common metalloids include Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), Tellurium (Te), and sometimes Polonium (Po).
How does metallic character change across a period?
From left to right, metallic character decreases and non-metallic character increases due to increasing nuclear charge.
How does metallic character change down a group?
From top to bottom, metallic character increases due to increasing atomic size and easier loss of electrons.
How do you find the period number of an element?
The period number is given by the principal quantum number (n) of the outermost shell in the electronic configuration.
How do you find the group number of s-block elements?
For s-block elements, if the outer configuration is ns¹ → Group 1, and if ns² → Group 2.
How do you find the group number of p-block elements?
Group number = 10 + (number of electrons in outermost s and p orbitals).
How do you find the group number of d-block elements?
Answer: Group number = (number of electrons in (n–1)d orbital) + (number of electrons in ns orbital).
How do you find the group number of f-block elements?
All f-block elements are placed in Group 3, regardless of their electron count.
What is the period number of f-block elements?
For lanthanides, n = 6 → Period 6; for actinides, n = 7 → Period 7
Important Chapter Links
To strengthen your understanding of this topic, you should also study Modern Periodic Law and Modern Periodic Table (Groups, Periods, Blocks) and Classification of Elements and Periodicity in Properties. It is equally important to revise Structure of Atom, as electronic configuration plays a key role in determining the position of elements. For better exam preparation, practice JEE Main and IMU CET Previous Year Questions (PYQs) and explore Class 11 Chemistry Chapter-wise Notes available on this website.