Atomic radius trend explains how the size of atoms changes across a period and down a group in the periodic table. As we move from left to right across a period, atomic radius generally decreases due to an increase in effective nuclear charge, which pulls electrons closer to the nucleus. On the other hand, as we move down a group, atomic radius increases because new electron shells are added, increasing the distance between the nucleus and outermost electrons. Understanding this variation is essential for explaining periodic properties and chemical behavior of elements.
- Variation of Atomic Radius in a Period
- Variation of Atomic Radius in a Second Period
- Anomalous Behaviour of Oxygen and Fluorine
- Why atomic radius of oxygen is slightly more than Nitrogen ?
- Variation of Atomic Radius in Third Period
- Variation of Atomic Radius in a Group
- FAQs – Frequently Asked Very Short Questions and Answers – Variation of Atomic Radius in a Period
Variation of Atomic Radius in a Period
In general, atomic radii decrease with an increase in atomic number when moving from left to right across a period in the periodic table.
Variation of Atomic Radius in a Second Period
In the second period, the atomic radii decrease from lithium (Li) to neon (Ne) through beryllium (Be), boron (B), carbon (C), nitrogen (N), oxygen (O), and fluorine (F), as shown in Table 1.
Table 1: Decrease in Atomic Radii in the Second Period
| Element (Atomic No.) | Nuclear Charge | Outer Electronic Configuration | Atomic Radius (pm) |
|---|---|---|---|
| ³Li | +3 | 2s¹ | 152 |
| ⁴Be | +4 | 2s² | 111 |
| ⁵B | +5 | 2s² 2p¹ | 88 |
| ⁶C | +6 | 2s² 2p² | 77 |
| ⁷N | +7 | 2s² 2p³ | 70 |
| ⁸O | +8 | 2s² 2p⁴ | 74 |
| ⁹F | +9 | 2s² 2p⁵ | 72 |
| ¹⁰Ne* | +10 | 2s² 2p⁶ | 160* |
** Note : For neon, the value given is the van der Waals radius (inert gas radius).
The variation in atomic radius with atomic number for the second period can be explained based on the increasing nuclear charge across the period.
As the atomic number increases from lithium to fluorine, the nuclear charge increases progressively by one unit. The additional electrons are added to the same principal shell (n = 2).
Screening effect is same across a period because inner electrons are not change across a period when new electron enters into valence shell. That is, since electrons in the same shell do not significantly shield or screen one another from the nucleus, the increased nuclear charge is not neutralized by the extra valence electrons.
As a result, the outer electrons are pulled closer to the nucleus by the greater effective nuclear charge, causing the atomic size to decrease steadily across the period.
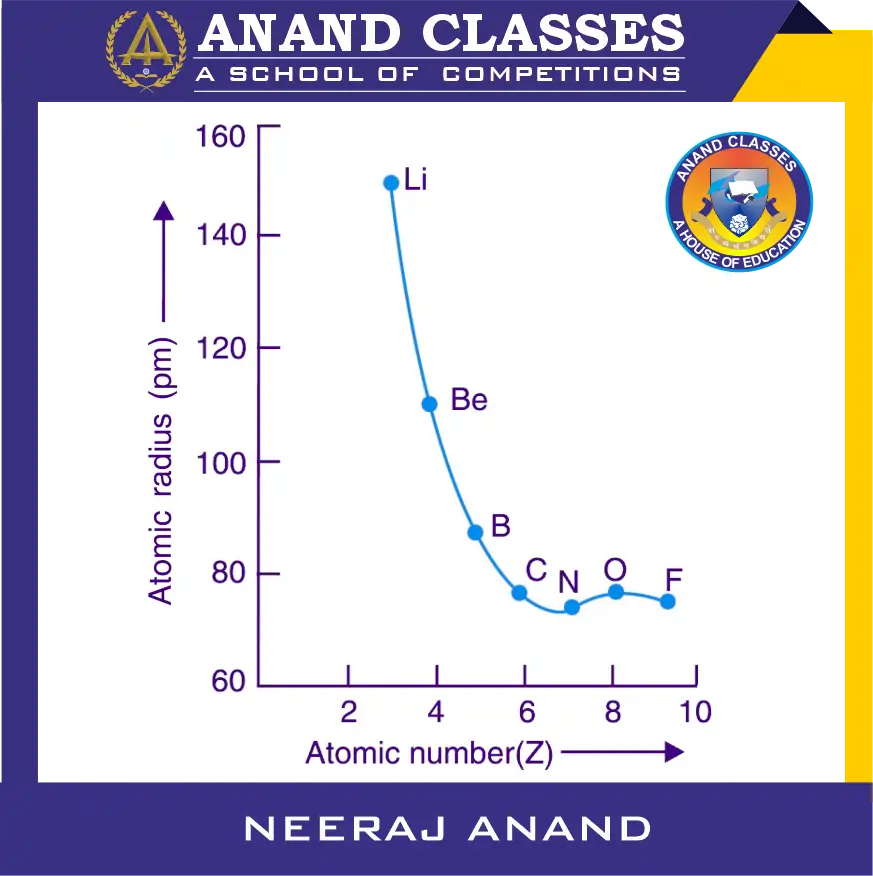
Anomalous Behaviour of Oxygen and Fluorine
Why atomic radius of oxygen is slightly more than Nitrogen ?
From Li to N, the atomic radius decreases regularly. However, after nitrogen, the atomic radius increases slightly for oxygen and then decreases again for fluorine in the above diagram.
- Nitrogen : All three 2p orbitals have one electron each (2s² 2px¹ 2py¹ 2pz¹).
- Oxygen : One of the 2p orbitals has two electrons (2s² 2pₓ² 2pᵧ¹ 2pz¹). The paired electrons in the same orbital experience increased interelectronic repulsion, which outweighs the effect of the increased nuclear charge, resulting in a slightly larger size than nitrogen.
- Fluorine : Two of the 2p orbitals have paired electrons (2s² 2pₓ² 2pᵧ2 2pz¹), but here the increased nuclear charge outweighs the repulsions, so the size decreases again compared to oxygen.
It is notable that both oxygen and fluorine have atomic sizes larger than nitrogen due to this interplay of nuclear charge and electron–electron repulsion.
Variation of Atomic Radius in Third Period
A similar trend is observed in the third period, as shown in Table.2.
Table 2: Decrease in Atomic Radii in the Third Period
| Element (Atomic No.) | Atomic Radius (pm) |
|---|---|
| ¹¹Na | 186 |
| ¹²Mg | 160 |
| ¹³Al | 143 |
| ¹⁴Si | 118 |
| ¹⁵P | 110 |
| ¹⁶S | 104 |
| ¹⁷Cl | 99 |
Thus, it may be concluded that in general (except for noble gases) :
- the alkali metals (Group 1) which are at the extreme left of the periodic table have the largest atomic radii in their respective periods because they have only one valence electron and weak nuclear attraction.
- the halogens (Group 17) which are present at the extreme right of the periodic table have much smaller atomic radii than alkali metals in the same period because their nuclei have more protons pulling electrons closer.
- the atomic size decreases along the period from left to right.
Variation of Atomic Radius in a Group
When we move down a group in the periodic table, the atomic radius of elements increases from top to bottom. This is due to reason that :
Increase in Nuclear Charge : As we go down a group, the atomic number (Z) increases. This means more protons are present in the nucleus, resulting in a greater nuclear charge. Normally, an increase in nuclear charge would pull the electrons closer to the nucleus, reducing the atomic size.
Addition of New Electron Shells (Principal Quantum Levels) : However, moving from one element to the next down the group also adds a new electron shell (new principal quantum number, n). This additional shell greatly increases the distance between the outermost electrons and the nucleus. Even though the number of electrons in the outermost shell remains the same, the distance of this shell from the nucleus increases with each step down the group.
Increase Screening (shielding) effect : Increase Screening (shielding) effect of inner electrons reduces the attraction between the nucleus and the valence electrons.
Dominance of Shell Addition Over Nuclear Attraction : The increase in the size of the electron cloud due to the addition of shells and increase screening effect is more significant than the increased nuclear attraction.
Therefore, the size of the atom increases as we move down the group.
Example.1 : Alkali Metals (Group 1)
| Element | Atomic Radius (pm) | Electronic Configuration | No. of Electron Shells |
|---|---|---|---|
| Li | 152 | 1s² 2s¹ | 2 |
| Na | 186 | 1s² 2s² 2p⁶ 3s¹ | 3 |
| K | 231 | 1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹ | 4 |
| Rb | 244 | [Kr] 5s¹ | 5 |
| Cs | 262 | [Xe] 6s¹ | 6 |
Observation :
Each step down the group from Li → Cs adds one new electron shell, causing a significant increase in atomic size despite the rise in nuclear charge.
Example.2 : Halogens (Group 17)
| Element | Atomic Radius (pm) | Electronic Configuration | No. of Electron Shells |
|---|---|---|---|
| F | 72 | 1s² 2s² 2p⁵ | 2 |
| Cl | 99 | 1s² 2s² 2p⁶ 3s² 3p⁵ | 3 |
| Br | 114 | [Ar] 4s² 3d¹⁰ 4p⁵ | 4 |
| I | 133 | [Kr] 5s² 4d¹⁰ 5p⁵ | 5 |
| At | 140 | [Xe] 6s² 4f¹⁴ 5d¹⁰ 6p⁵ | 6 |
Observation:
- Similar to alkali metals, new shells are added down the group, increasing the atomic size.
- The increase is less steep compared to Group 1 due to stronger nuclear attraction in nonmetals.
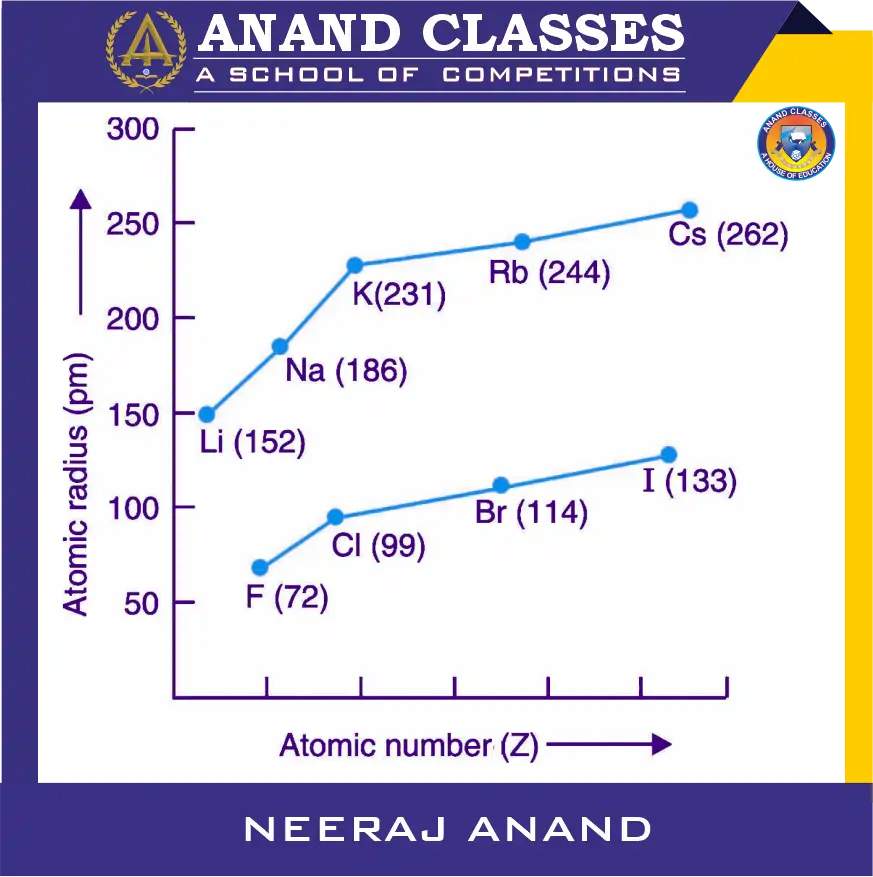
Graphical Representation
When plotted, the atomic radius vs. element position in a group shows a steady upward trend from top to bottom. This confirms that addition of shells is the dominant factor in determining atomic size within a group.
FAQs – Frequently Asked Very Short Questions and Answers – Variation of Atomic Radius in a Period
Why does atomic radius decrease across a period?
As we move from left to right in a period, the number of protons in the nucleus increases. This leads to a stronger nuclear charge pulling electrons closer to the nucleus. Since electrons are added to the same energy level, shielding remains almost constant, resulting in a decrease in atomic size.
Which element in a period has the smallest atomic radius?
The halogens at the end of the period usually has the smallest atomic radius because it has the highest effective nuclear charge among the elements in that period.
Does the shielding effect change across a period?
Only slightly. Electrons are added to the same shell, so shielding effect does not increase significantly. The increase in nuclear charge dominates, causing the atomic radius to shrink.
Are there any exceptions to the trend of decreasing atomic radius?
Yes, slight anomalies exist due to electron-electron repulsion in half-filled and fully filled orbitals. For example, oxygen has a slightly larger radius than nitrogen because of extra electron repulsion in the paired electrons of the 2p orbital.
Why does atomic radius increase down a group?
As we move down a group, a new electron shell (principal quantum level) is added to the atom for each successive element. This increases the distance between the outermost electrons and the nucleus, making the atom larger despite the increase in nuclear charge.
Does increasing nuclear charge not decrease atomic size?
Normally, greater nuclear charge would pull electrons closer, reducing atomic size. However, in a group, the effect of additional electron shells outweighs the increased nuclear attraction, leading to a larger atomic radius.
How does the number of valence electrons change down a group?
The number of valence electrons remains the same for all elements in a group. For example, alkali metals always have one valence electron, and halogens always have seven, regardless of their position down the group.
Which group has the largest atomic radii — alkali metals or halogens?
Alkali metals (Group 1) have much larger atomic radii compared to halogens (Group 17) in the same period because alkali metals have fewer protons and weaker nuclear attraction for their valence electrons.
What is the trend in atomic radius among alkali metals?
It increases from lithium (Li) to caesium (Cs) because each element has an additional electron shell compared to the one above it.
What is the trend in atomic radius among halogens?
It increases from fluorine (F) to astatine (At) for the same reason — addition of extra electron shells down the group.
How does shielding effect relate to atomic radius in a group?
As more inner electron shells are added, the shielding effect increases, reducing the effective nuclear charge experienced by the valence electrons, which allows them to be farther from the nucleus, increasing atomic radius.
Important Chapter Interlinks
This section provides a complete and interconnected study of Classification of Elements and Periodicity in Properties, starting with detailed theory and notes for Class 11 Chemistry to build a strong conceptual foundation. You can explore atomic radius and its types including covalent, van der Waals, metallic, and ionic radii to understand periodic trends in atomic size. It also includes Screening Effect (Shielding Effect) : Calculation of Effective or Reduced Nuclear Charge (Slater’s Rules), which explains how inner electrons reduce the nuclear attraction on outer electrons and influence periodic trends. The causes of periodicity explain why elements show repeating properties based on electronic configuration, which is further supported by the modern periodic law and structure of the modern periodic table including groups, periods, and blocks for elements even beyond atomic number 100. The historical development is covered through Mendeleev’s periodic law and table, leading to the modern classification of elements into s, p, d, and f blocks with prediction of period, group, and block. To strengthen exam preparation, you can practice JEE Main PYQs, IMU CET PYQs and Merchant Navy sponsorship exam MCQs, and other previous year questions with solutions, along with solved examples, conceptual questions, and practice problems on the modern periodic table. Additionally, complete study material, mock tests, and guidance are provided under Anand Classes Chemistry notes, along with expert support from Er Neeraj Anand, making this section a comprehensive resource for competitive exam preparation.
