The Modern Periodic Law and Modern Periodic Table provide a systematic and scientific arrangement of elements based on their atomic numbers. Unlike earlier classifications, the modern periodic table explains periodicity in terms of electronic configuration, making it more accurate and reliable. Elements are arranged in rows called periods and columns called groups, and are further divided into blocks such as s-block, p-block, d-block, and f-block. This classification helps in understanding trends in properties, predicting chemical behavior, and forming a strong foundation for advanced chemistry concepts. Modern periodic table extends beyond atomic number 100 with the inclusion of superheavy elements that are artificially synthesized in laboratories. According to the Modern Periodic Law, these elements are arranged based on their atomic numbers and electronic configurations, fitting systematically into groups, periods, and blocks of the periodic table. Most of these elements belong to the f-block and d-block regions and are highly unstable with very short lifetimes. Despite their instability, their placement in the modern periodic table helps in understanding periodic trends, nuclear stability, and the expansion of chemical knowledge.
- Moseley’s Experiment and Observation
- Modern Periodic Law and Modern Periodic Table (According to Atomic Number)
- The Long Form Of The Periodic Table (Bohr’s Table)
- Seven Periods In a Periodic Table
- Electronic Configurations of Elements in Periods
- Eighteen Groups In a Periodic Table
- Nomenclature System for the Elements with Atomic Numbers More than 100
- IUPAC Systematic (Temporary) Naming Rules
- Elements Z = 101 to 118 with Temporary & Official Names
- Frequently Asked Questions (FAQs) and Answers Based on Modern Periodic Table

Moseley’s Experiment and Observation
In 1913, Moseley, a young English physicist discovered the relationship between X-ray spectra and the atomic number of the elements. When high energy electrons were focused on a target made of the elements under study, X-rays were generated. He studied the frequencies of X-rays emitted from the elements and observed that the frequency (ν) of the prominent X-rays emitted by an element was proportional to the atomic number and not to the atomic weight. He gave a general equation :
$$\sqrt{\nu} = a(Z – b)$$
where ν is the frequency of the emitted X-ray and a and b are constants that are same for all the elements. A plot of $\sqrt{\nu}$ versus atomic number (Z) gives a straight line.
Henry Moseley conducted an experiment involving high-velocity electrons directed at metallic targets. He observed:
The square root of the frequency (ν) of X-rays emitted is proportional to the atomic number (Z) of the element.
$$\sqrt{\nu} = a(Z – b)$$
This led Moseley to conclude that atomic number and not atomic weight is the fundamental property of the atoms. He, therefore, suggested that atomic number instead of atomic weight should be the basis of classification of elements.
The acceptance of atomic number, as the important characteristic of an atom, led to the modern periodic law.
Modern Periodic Law and Modern Periodic Table (According to Atomic Number)
Modern periodic law may be stated as the physical and chemical properties of the elements are periodic functions of their atomic numbers.
This law forms the basis of the Modern or Long Form of the Periodic Table.
This law implies that the physical and chemical properties of the elements depend on atomic number and this dependency shows periodicity. Consequently, when the elements are arranged in the order of their increasing atomic numbers, it is observed that the elements of similar properties recur at regular intervals or periodically. As a result of this, the elements fall in certain groups and lead to an arrangement called the periodic table.
With the modern periodic law as the guiding principle and the modern theory of atomic structure, many new forms of the periodic table have been proposed from time to time. Some forms emphasise chemical reactions and valence, whereas other forms stress the electronic configurations of elements. However, the general plan of the table has remained the same as proposed by Mendeleev. Furthermore, it is now recognized that the periodic law is essentially the consequence of the periodic variation in electronic configurations of atoms, which determine the physical and chemical properties of the elements and their compounds. To read Mendeleev’s Periodic Law and Mendeleev’s Periodic Table Click the given link.
The Long Form Of The Periodic Table (Bohr’s Table)
The long form of the periodic table is designed according to the Modern Periodic Law and is also referred to as Bohr’s Table, as it reflects Bohr’s theory of electron arrangement in atoms. Modern periodic table is also called long form of the periodic table or Bohr’s table.
The most widely used periodic table these days is ‘The Long Form of the Periodic Table’. This is constructed on the basis of repeating electronic configuration of the atoms when the elements are arranged in the order of increasing atomic numbers.
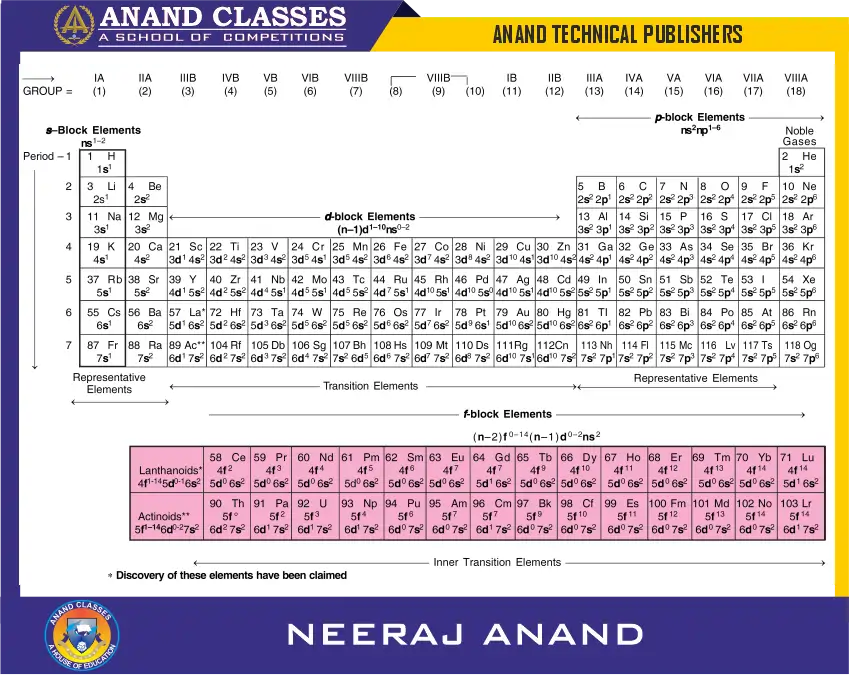
The long form of the periodic table consists of horizontal rows called periods and vertical columns called groups. There are altogether seven periods. These are numbered as 1, 2, 3, 4, 5, 6 and 7. The first period contains 2 elements and the subsequent periods consist of 8, 8, 18, 18, 32 and 32 elements respectively. Fourteen elements of both sixth and seventh periods (called lanthanoids and actinoids respectively) are placed in separate columns at the bottom of the periodic table.
There are eighteen groups in the long form of the periodic table. Earlier, these groups were designated as IA, IB, IIA, IIB…. VIII or zero groups. According to new recommendations of the International Union of Pure and Applied Chemistry (IUPAC), the groups are numbered from 1 to 18.
Seven Periods In a Periodic Table
A horizontal row in the periodic table is called period.
In terms of electronic structure of the atom, a period constitutes a series of elements whose atoms have the same number of electron shells i.e. principal quantum number (n). Thus, the period indicates the value of n for the outermost or valence shell of the atom of the element. Each successive period in the periodic table is associated with the filling of the next higher principal energy level (n=1, n=2, n=3, etc.) There are seven periods and each period starts with a different principal quantum number.
It can be seen that the number of elements in each period is twice the number of atomic orbitals available in the energy level that is being filled.
Electronic Configurations of Elements in Periods
Electronic configurations in periods follow a systematic filling of energy subshells, where the period number corresponds to the principal quantum number (n) of the valence shell. As you move left to right, electrons are added to the same energy level (s and p subshells).
Elements of First Period
The first period corresponding to n = 1 is unique because it contains only two elements. This is not surprising because first energy shell (K) has only one orbital (i.e. 1s) which can accommodate only two electrons. This means that there can be only two elements in which one and two electrons are present in first energy level. The first period contains hydrogen (1s¹) and helium (1s²).
Elements of Second Period
The second period contains 8 elements because for n = 2, there are four orbitals (one 2s and three 2p) in second energy shell (L). In all, these four orbitals have a capacity of eight electrons and, therefore, second period has eight elements in it. It starts with lithium (Z = 3) in which one electron enters the 2s-orbital. The period ends with neon (Z = 10) in which the second shell is complete (2s² 2p⁶).
Elements of Third Period
In third period corresponding to n = 3, there are nine orbitals : one 3s, three 3p and five 3d. However, we know from energy level diagram for multielectron atoms (Unit 2) that 3d-orbitals are higher in energy than 4s-orbitals. Consequently, 3d-orbitals are filled after filling 4s-orbital. Hence, this period involves the filling of only four orbitals (3s and 3p) and contains eight elements from sodium (Z = 11) to argon (Z =18).
Elements of Fourth Period
The fourth period corresponding to n = 4 involves the filling of one 4s-and three 4p-orbitals (4d and 4f orbitals are higher in energy than 5s-orbitals and are filled later). In between 4s-and 4p-orbitals, five 3d-orbitals are filled which have energies in between these orbitals. Thus, in all nine orbitals are to be filled and, therefore, there are eighteen elements in fourth period from potassium (Z = 19) to krypton (Z = 36). In this period, we come across elements which involve the filling of 3d orbitals. These are known as transition series of elements. This starts from scandium (Z = 21) which has electronic configuration 3d¹ 4s² and ends at zinc (Z = 30) with 3d orbitals completely filled having electronic configuration 3d¹⁰ 4s².
Elements of Fifth Period
The fifth period, like the fourth period also consists of 18 elements. It begins with rubidium (Z = 37) with filling of 5s-orbital and ends at xenon (Z = 54) with the filling up of the 5p-orbitals. This period also contains ten elements of 4d transition series starting at yttrium (Z = 39).
Elements of Sixth Period
The sixth period contains 32 elements (Z = 55 to 86) and successive electrons enter into 6s, 4f, 5d and 6p-orbitals, in that order. It starts with cesium (Z = 55) and ends at radon (Z = 86). In addition to ten elements of 5d transition series, this period contains 14 elements which involve filling up of 4f orbitals beginning from cerium (Z = 58) to lutetium (Z = 71). The series of elements are called inner transition series or lanthanoid series.
Elements of Seventh Period
The seventh period, is similar to the sixth period with successive filling up of the 7s, 5f, 6d and 7p orbitals and includes most of the man made radio-active elements. This period ends at the element with atomic number 118, which would belong to the noble gas family. Some of the elements of this period have been recently established and named. This period also contains 14 elements which involve the filling of 5f orbitals starting from actinium (Z = 89). This is also called 5f– inner transition series or actinoid series.
This also provides a theoretical justification for periodicity occurring at regular intervals of 2, 8, 8, 18, 18 and 32. These numbers i.e., 2, 8, 18 and 32 are also called magic numbers. The number of elements and the corresponding orbitals being filled are given in Table below.
Table. Number of elements (corresponding to electrons) in each period.
| Period (n) | Orbitals being filled up | Number of electrons or elements in the period |
|---|---|---|
| First (1) | 1s | 2 = 2 |
| Second (2) | 2s 2p | 2 + 6 = 8 |
| Third (3) | 3s 3p | 2 + 6 = 8 |
| Fourth (4) | 4s 3d 4p | 2 + 10 + 6 = 18 |
| Fifth (5) | 5s 4d 5p | 2 + 10 + 6 = 18 |
| Sixth (6) | 6s 4f 5d 6p | 2 + 14 + 10 + 6 = 32 |
| Seventh (7) | 7s 5f 6d 7p | 2 + 14 + 10 + 6 = 32 |
The first three periods containing 2, 8 and 8 elements respectively are called short periods, the next three periods containing 18, 18 and 32 elements respectively are called long periods.
Eighteen Groups In a Periodic Table
A vertical column in the periodic table is known as group.
In terms of electronic structure of the atom, a group constitutes a series of elements whose atoms have the same outermost electronic configurations. There are 18 groups in the long form of the periodic table. According to the new recommendations of International Union of Pure and Applied Chemistry (IUPAC), the groups are numbered from 1 to 18. Previously, these were numbered only from I to VIII as A and B groups. Both the systems of numbering the groups is given in the periodic table. However, the old convention is still used in many books.
The first two groups on the extreme left and last six groups on the extreme right involve the filling of s-and p-orbitals, respectively. These groups represent the main groups of the periodic table and are numbered as 1, 2, 13, 14, 15, 16, 17 and 18 corresponding to general configurations of ns¹, ns², ns² np¹, ns² np², ns² np³, ns² np⁴, ns² np⁵, ns² np⁶, respectively (in the old system these were denoted as IA, IIA, IIIA, IVA, VA, VIA, VIIA and VIIIA (or zero) respectively corresponding to general configurations of ns¹, ns², ns² np¹,……….ns² np⁶ respectively. The elements present in these groups are known as normal or representative elements.
The first three periods containing 2, 8 and 8 electrons involve the filling of only s-and p-subshells. But the fourth period involves the filling of 3d-subshells also and this results into ten more groups. These ten groups lie in between first two and last six groups, (see periodic table) i.e., between group 2 (IIA) and group 13 (IIIA) because 3d-orbitals are filled after 4s-orbitals but before 4p-orbitals. These are numbered from 3 to 12 (in the old system these were usually labelled as B groups i.e., IB, IIB, IIIB,…etc.). The elements present in these groups are called transition elements. The name is derived from the fact that they represent transition (change) in character from reactive metals (elements of groups 1 and 2) on one side to the non-metals (elements of group 13 to 18) on the other side.
There are two more rows at the bottom of the periodic table. These rows consist of fourteen elements after lanthanum (Z = 57) and fourteen elements which follow actinium (Z = 89). These are placed separately in the periodic table to save space and avoid undue sidewise expansion of the periodic table. This arrangement also helps in keeping elements with similar properties in a single column. The elements in the first row, starting from cerium are called lanthanoids (or lanthanides) and the elements present in the second row starting from thorium are called actinoids (or actinides). These lanthanoids and actinoids together are called inner transition elements or rare earth metals and these are built up by filling of f-orbitals.
Nomenclature System for the Elements with Atomic Numbers More than 100
Today, we have elements upto 118. These elements were earlier named traditionally by its discoverer or discoverers. However, in recent years, disputes regarding the original discoveries of some elements of atomic number 104 and above have arisen. This is because the new elements with very high atomic numbers are very unstable and only minute quantities of elements are obtained. This has led to controversy in deciding the claims of more than one scientist for the discovery and names assigned by them for the new element discovered.
For example, both American and Soviet scientists claimed credit for discovery of element 104. The Americans named it Rutherfordium (symbol Rf) while Soviets named it Kurchatovium (symbol Ku). To overcome this problem, the IUPAC in 1997 gave the approved official names for elements with atomic numbers 104 to 109. The IUPAC has also recommended a systematic nomenclature for naming the new elements until their names are officially recognized. This nomenclature is based on the Latin words for their numbers.
IUPAC Systematic (Temporary) Naming Rules
The temporary name for any element with Z > 100 is constructed directly from its digits using specific Latin/Greek-derived numerical roots, followed by the suffix “-ium”.
Step-by-step rule:
- Break the atomic number into its individual digits.
Example: 115 → digits are 1, 1, 5. - Replace each digit with the corresponding prefix from the table below (in order):
| Digit | Prefix |
|---|---|
| 0 | nil |
| 1 | un |
| 2 | bi |
| 3 | tri |
| 4 | quad |
| 5 | pent |
| 6 | hex |
| 7 | sept |
| 8 | oct |
| 9 | enn |
- Concatenate these prefixes in the same order as the digits and add the suffix “-ium”.
So, 115 → un (1) + un (1) + pent (5) → ununpentium. - Apply vowel contraction if necessary:
If concatenation leads to two identical vowels in a row, typically one is dropped to make pronunciation smoother.- E.g., “bi + un” would be biun (not biiun).
- Rare cases are handled to avoid awkward double vowels.
- Symbol formation:
The symbol is formed by taking the first letter of each numerical prefix, capitalizing the first, and using the next letters in lowercase if needed to avoid ambiguity.- Example: ununpentium → prefixes are un, un, pent → symbol Uup (U-u-p).
- Final name ends with “-ium.”
Every systematic temporary name ends in “-ium” regardless of the element’s chemical character.
Examples of Temporary Names
| Atomic Number | Systematic Temporary Name | Symbol (temporary) |
|---|---|---|
| 101 | unnilunium | Unu |
| 102 | unnilbium | Unb |
| 103 | unniltrium | Unt |
| 104 | unnilquadium | Unq |
| 105 | unnilpentium | Unp |
| 106 | unnilhexium | Unh |
| 107 | unnilseptium | Uns |
| 108 | unniloctium | Uno |
| 109 | unnilennium | Une |
| 110 | ununnilium | Uun |
| 111 | unununium | Uuu |
| 112 | ununbium | Uub |
| 113 | ununtrium | Uut |
| 114 | ununquadium | Uuq |
| 115 | ununpentium | Uup |
| 116 | ununhexium | Uuh |
| 117 | ununseptium | Uus |
| 118 | ununoctium | Uuo |
(Note: These were temporary names used before official names were ratified.)
Transition to Permanent Official Names
- Once an element’s discovery was confirmed through peer-reviewed evidence and vetted by IUPAC/IUPAP joint working groups, a permanent name was proposed, typically based on:
- A scientist (e.g., Seaborgium, after Glenn T. Seaborg),
- A place (e.g., Darmstadtium, after Darmstadt),
- A mythological concept, or
- A property (less common for superheavy).
- After a period of public review, the name becomes official and replaces the temporary systematic name. Symbols likewise are updated to the permanent two-letter (or sometimes one-letter) symbols.
Elements Z = 101 to 118 with Temporary & Official Names
| Z | Temporary Name | Temp. Symbol | Official Name | Official Symbol |
|---|---|---|---|---|
| 101 | Unnilunium | Unu | Mendelevium | Md |
| 102 | Unnilbium | Unb | Nobelium | No |
| 103 | Unniltrium | Unt | Lawrencium | Lr |
| 104 | Unnilquadium | Unq | Rutherfordium | Rf |
| 105 | Unnilpentium | Unp | Dubnium | Db |
| 106 | Unnilhexium | Unh | Seaborgium | Sg |
| 107 | Unnilseptium | Uns | Bohrium | Bh |
| 108 | Unniloctium | Uno | Hassium | Hs |
| 109 | Unnilennium | Une | Meitnerium | Mt |
| 110 | Ununnilium | Uun | Darmstadtium | Ds |
| 111 | Unununium | Uuu | Roentgenium | Rg |
| 112 | Ununbium | Uub | Copernicium | Cn |
| 113 | Ununtrium | Uut | Nihonium | Nh |
| 114 | Ununquadium | Uuq | Flerovium | Fl |
| 115 | Ununpentium | Uup | Moscovium | Mc |
| 116 | Ununhexium | Uuh | Livermorium | Lv |
| 117 | Ununseptium | Uus | Tennessine | Ts |
| 118 | Ununoctium | Uuo | Oganesson | Og |
Frequently Asked Questions (FAQs) and Answers Based on Modern Periodic Table
What is the Modern Periodic Law?
The Modern Periodic Law states that “The physical and chemical properties of elements are periodic functions of their atomic numbers.” This means the properties of elements repeat at regular intervals when arranged by increasing atomic number.
Who gave the Modern Periodic Law and why?
Henry Moseley gave the Modern Periodic Law in 1913 after discovering that atomic number is a more fundamental property than atomic mass. His work corrected the anomalies in Mendeleev’s periodic table.
How many periods are there in the Modern Periodic Table?
There are seven periods (horizontal rows) in the Modern Periodic Table. Each period corresponds to the number of electron shells in the elements.
How many groups are present in the Modern Periodic Table?
The Modern Periodic Table has 18 vertical columns, known as groups, which classify elements with similar outermost electron configurations.
What are the 4 blocks in the Modern Periodic Table?
The periodic table is divided into four blocks based on the subshell being filled:
s-block (Groups 1 and 2)
p-block (Groups 13 to 18)
d-block (Groups 3 to 12)
f-block (Lanthanides and Actinides)
What are representative elements?
Elements of the s- and p-blocks (except noble gases and hydrogen) are called representative elements. They exhibit variable valency and typical chemical properties.
What are transition elements?
d-block elements (Groups 3–12) are called transition elements. They form colored compounds, have variable oxidation states, and act as good catalysts.
What are inner transition elements?
f-block elements (lanthanides and actinides) are called inner transition elements. They are placed separately at the bottom of the periodic table to keep it compact.
Which is the shortest and the longest period in the periodic table?
The 1st period is the shortest, with only 2 elements (H and He).
The 6th period is the longest, containing 32 elements, including lanthanides.
Why do elements with atomic number greater than 100 need special nomenclature?
Because many of these superheavy elements are synthetic and discovered in different labs, their naming often caused disputes. To avoid confusion, IUPAC introduced a systematic temporary naming system based on atomic numbers until permanent names are approved.
What is the general rule for naming elements with Z > 100?
Each digit of the atomic number is replaced by a prefix (derived from Latin/Greek), combined in order, followed by the suffix “-ium.”
How is the symbol of a temporary name decided?
The symbol is formed from the first letters of the prefixes (capitalizing the first), e.g., ununpentium (115) → Uup.
Give an example of temporary and permanent names.
Z = 114 → Temporary: ununquadium (Uuq) → Permanent: Flerovium (Fl).
Z = 118 → Temporary: ununoctium (Uuo) → Permanent: Oganesson (Og).
Why do some heavier elements show awkward names like “unununium”?
Because their atomic numbers have repeating digits, leading to repeating prefixes (e.g., 111 → un-un-un → unununium).
Who decides the permanent name of an element?
Permanent names are decided and approved by IUPAC after confirming the element’s discovery. The name usually honors a scientist, place, or property.
What is the current range of officially named elements?
As of now, elements are named up to Z = 118 (Oganesson, Og). Beyond that, elements are still under research and have no confirmed permanent names.
Why is the periodic table important for JEE and NEET?
The periodic table helps students predict element properties, understand periodic trends, and classify elements based on atomic structure. It forms the basis for many questions in JEE, NEET, and Class 11 Chemistry.
Summary
For a complete understanding of this topic, students should also explore other important Class 11 Chemistry chapters available on this website. You can study Classification of Elements and Periodicity in Properties, Structure of Atom to understand atomic properties, Chemical Bonding and Molecular Structure for bonding concepts, and Thermodynamics and Equilibrium for reaction behavior and energy changes. It is also recommended to practice JEE Main Previous Year Questions (PYQs) and revise from chapter-wise notes to strengthen problem-solving skills. Exploring these related topics will help you connect concepts and achieve better results in exams like JEE and NEET.
Buy Study Material & Join Our Coaching
For premium study materials specially designed for JEE, NEET, NDA, and CBSE/ICSE Classes, visit our official study material portal:
https://publishers.anandclasses.co.in/
To enroll in our offline or online coaching programs, visit our coaching center website:
https://anandclasses.co.in/
Call us directly at: +91-94631-38669
WhatsApp Us Instantly
Need quick assistance or want to inquire about classes and materials?
Click below to chat instantly on WhatsApp:
Chat on WhatsApp
Watch Video Lectures
Get access to high-quality video lessons, concept explainers, and revision tips by subscribing to our official YouTube channel:
Neeraj Anand Classes – YouTube Channel